That was enough to get slapped down by the Federal Trade Commission and rightly so. Though the U.S. Food and Drug Administration has been forced to allow all manner of suspect claims in the food and supplement industry due to President Clinton's 1994 Dietary Supplement Health and Education Law exempting supplement hucksters from any real oversight, overtly lying to is another animal.
Literally. Just like British company Diageo had to walk back suggestions they were producing healthier vodka because Smirnoff switched to non-GMO corn - the Treasury Department's Alcohol and Tobacco Tax and Trade Bureau is not crippled when it comes to stopping nonsense the way FDA is - you can't claim in promotional materials that your magic birth juice will reverse macular degeneration, Parkinson’s Disease, and even cure autism.
If you do, the FTC will put a hex on you that magic potions can't cure. They can get an injunction against you if they have evidence you are defrauding consumers. Which this clearly was, for over $3 million dollars.
Stem cell therapy will some day be able to do great things. It is certainly more advanced than probiotic nonsense, which gets away with a lot by noting in small print that FDA has not validated anything they claim. Unfortunately so much promise leaves the door open for charlatans to exploit people with hope and money.
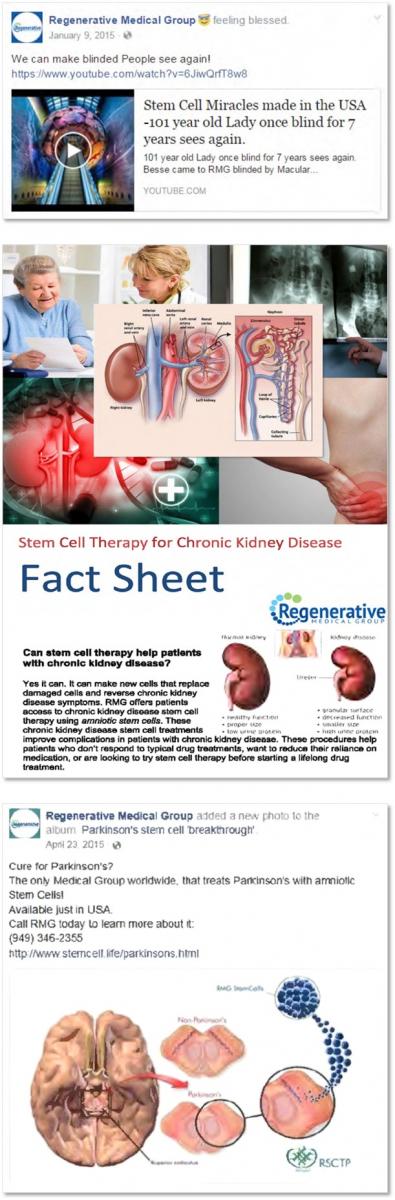
Graphic courtesy of FTC.




Comments