Biologists have induced one species of flatworm to grow heads and brains characteristic of another species of flatworm without altering the genomic sequence. The work reveals physiological circuits as a new kind of epigenetics - information existing outside of genomic sequence - that determines large-scale anatomy.
The finding that head shape is not hard-wired by the genome but can be overridden by manipulating electrical synapses in the body suggests that differences in species could be determined in part by the activity of bioelectrical networks. The discovery could help improve understanding of birth defects and regeneration by revealing a new pathway for controlling complex pattern formation. It has long been known that neural networks exploit bioelectric synapses to store and re-write information in the brain.
"It is commonly thought that the sequence and structure of chromatin - material that makes up chromosomes - determine the shape of an organism, but these results show that the function of physiological networks can override the species-specific default anatomy," says the paper's senior and corresponding author Michael Levin, Ph.D., of Tufts University. "By modulating the connectivity of cells via electrical synapses, we were able to derive head morphology and brain patterning belonging to a completely different species from an animal with a normal genome."
Knowing how shape is determined and how to influence it is important because biologists could use that knowledge, for example, to fix birth defects or cause new biological structures to grow after an injury, adds Levin. "These findings raise significant questions about how genes and bioelectric networks interact to build complex body structures," he says.
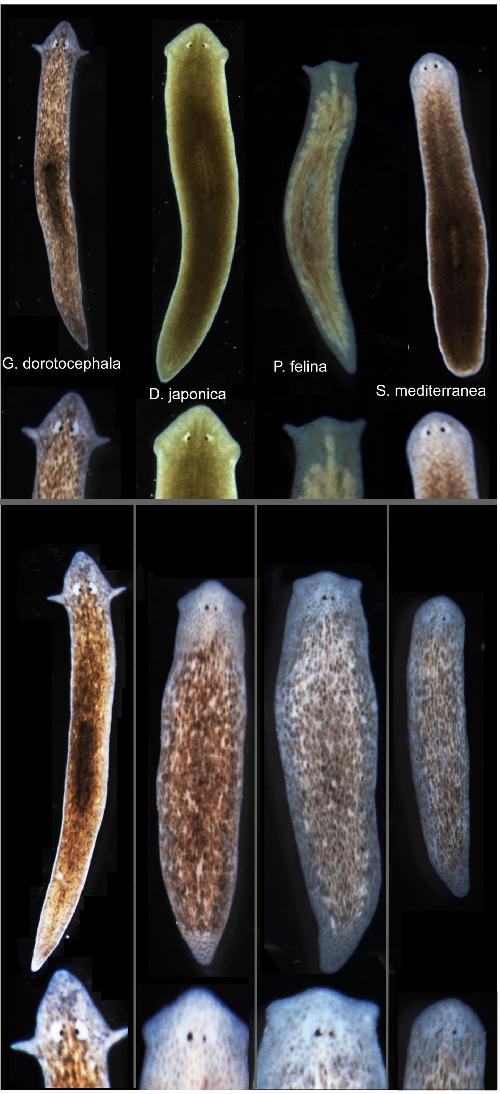
Working with Girardia dorotocephala - free-living planarian flatworms, which have remarkable regenerative capacity - researchers were able to induce the development of different species-specific head shapes by interrupting gap junctions, which are protein channels that enable cells to communicate with each other by passing electrical signals back and forth. The changes were more than skin deep; they included not only the overall shape of the head but also the shape of the brain and the distribution of the worm's adult stem cells.
The ease with which a particular shape could be coaxed from a G. dorotocephala worm was proportional to the proximity of the target worm on the evolutionary timeline. The closer the two species were related, the easier it was to effect the change. This observation strengthens the connection to evolutionary history, suggesting that modulation of physiological circuits may be one more tool exploited by evolution to alter animal body plans.
However, unlike the Levin lab's previous work, in which a different species of planaria could be permanently altered to a two-headed morphology, this shape change was only temporary. Weeks after the planaria completed regeneration to the other species' head shapes, the worms once again began remodeling and re-acquired their original head morphology. Additional research is needed to determine how this occurs. The authors also presented a computational model that explains how changes in cell-to-cell communication can give rise to the diverse shape types.
First author on the paper was Tufts undergraduate Maya Emmons-Bell, a senior majoring in biology. "We've demonstrated that the electrical connections between cells provide important information for species-specific patterning of the head during regeneration in planarian flatworms," she says. "This kind of information will be crucial for advances in regenerative medicine, as well as a better understanding of evolutionary biology. As an undergraduate, it's been an extraordinary experience to conduct and author important research with leading biologists."
Published in the International Journal of Molecular Sciences.





Comments