But the story of water gets a lot more scientifically interesting the deeper you go. Water actually exists in two different forms, called isomers, at the molecular level.
They have almost identical physical properties, you can't tell the difference, but chemists can tell them apart by the relative orientation of the nuclear spins of the two hydrogen atoms. They are called ortho- or para-water depending on whether the spins are aligned in the same or opposite direction.
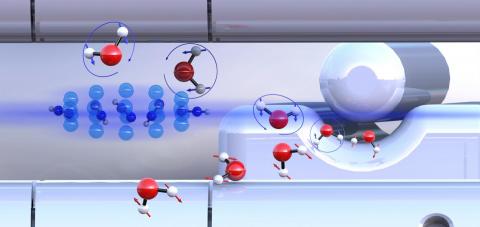
Pre-sorted ortho-water and para-water molecules with differently oriented nuclear spins (blue or red arrows) react with diazenylium ions (center left) at different speeds. Illustration: University of Basel
How are they really different?
In a recent experiment, separation was made possible by a method based on electric fields. The researchers were able to initiate controlled reactions between the "pre-sorted" water isomers and ultracold diazenylium ions ("protonated nitrogen") held in a trap. During this process, a diazenylium ion transfers its proton to a water molecule.
Fun fact: This reaction is also observed in the chemistry of interstellar space.
In their experiments, the researchers worked with molecules at very low temperatures close to the absolute zero point (about -273°C). These are ideal conditions to precisely prepare individual quantum states and define the energy content of the molecules, and to cause a controlled reaction between them.
They found that para-water reacts about 25% faster than ortho-water, which can be explained in terms of the nuclear spin also influencing the rotation of the water molecules. As a result, different attractive forces act between the reaction partners. Para-water is able to attract its reaction partner more strongly than the ortho-form, which leads to an increased chemical reactivity.
Look for magic water proponents to start claiming they are selling different types of water and that para-water is superior and you should pay more. Don't be fooled. Outside chemistry labs or interstellar space, it is even less meaningful than alkaline water.





Comments