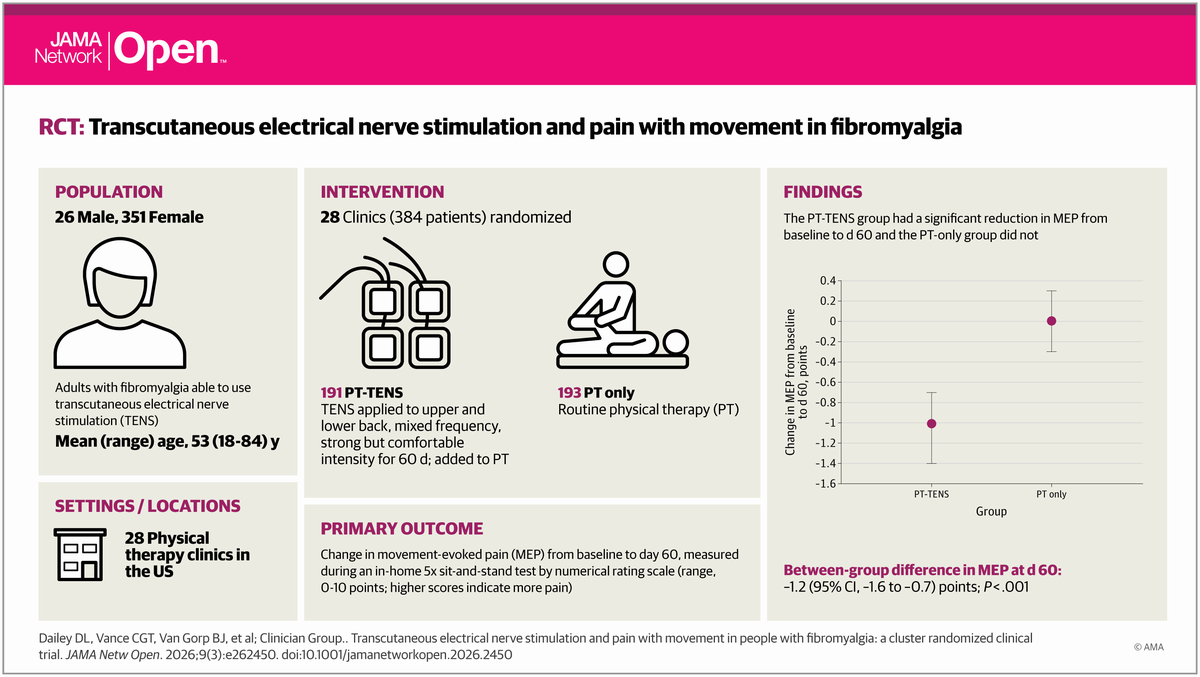
 Pilot Study: Fibromyalgia Fatigue Improved By TENS Therapy
Pilot Study: Fibromyalgia Fatigue Improved By TENS TherapyFibromyalgia is the term for a poorly-understood condition where people experience pain and fatigue...
 High Meat Consumption Linked To Lower Dementia Risk
High Meat Consumption Linked To Lower Dementia RiskOlder people who eat large amounts of meat have a lower risk of dementia and cognitive decline...
 Long Before The Inca Colonized Peru, Natives Had A Thriving Trade Network
Long Before The Inca Colonized Peru, Natives Had A Thriving Trade NetworkA new DNA analysis reveals that long before the Incan Empire took over Peru, animals were...
 Mesolithic People Had Meals With More Tradition Than You Thought
Mesolithic People Had Meals With More Tradition Than You ThoughtThe common imagery of prehistoric people is either rooting through dirt for grubs and picking berries...