Causes, Effects and Management of ‘Blue Green Algae’ Cyanobacteria and their Harmful Algal Blooms in Australian Recreation and Drinking Water with Two Ballina NSW Case Studies
Posted 21st June 2014 on Global MND/ALS Awareness Day in memory of MumCyanobacteria are one of the Earth’s oldest life forms believed to be responsible for colonising the planet and making it habitable for other life forms by reshaping the biosphere 3.5 billion years ago. Cyanobacteria otherwise known as ‘blue-green algae’ can reproduce rapidly under the right conditions to form harmful algal blooms (HABs) often referred to as cyanoHABs. CyanoHABs can take multiple shapes, forms and sizes; they may occur as single cells floating free within the water column or in groups as colonies or as straight, coiled or twisted filaments and form films or ‘mats’ that are clearly visible to the human eye.

Photo taken from Paerl&Paul 2012
CyanoHABs are only categorised as harmful if their blooms cause harm to other organisms. This harm may be caused by cyanoHABs blocking another organism’s light source, adversely obstructing or chemically disrupting their environment, outcompeting with them for available nutrients and resources or by producing harmful toxins called cyanotoxins, which can damage the health of other organisms in a variety of ways. The majority of cyanotoxins can be regarded as secondary metabolites that are not utilised by the organism’s primary metabolism and which can be classified into two main groups, those that cause acute lethal poisoning to other organisms which are the neurotoxins and hepatotoxins and those that are not lethal to other organisms but demonstrate selective bioactivity which are the cytotoxins (Whitton and Potts, 2000).
Cyanobacteria are often referred to as ‘blue-green algae’ however this is a misnomer as they belong to the group of organisms called prokaryotes which includes bacteria but does not include algae which are eukaryotes. Prokaryotes do not possess a true cell nucleus and other membrane bound cell compartments like mitochondria and chloroplasts that are found in the eukaryotes, such as animals, plants, fungi and protists. Cyanobacteria do however possess internal gas vacuoles and these enable them to be highly motile and competitive against less motile zooplankton and phytoplankton competitors especially within the water column where they are able to take advantage of nutrients available in different locations,temperatures and depths at different times. Cyanobacteria are capable of photosynthesis across a broad spectrum of ultra violet light and during adverse environmental conditions they can form dormant states such as thick-walled heterocytes containing the enzyme nitrogenase responsible for nitrogen fixation or cyanobacterial spores and akinetes that may re-emerge in the future under more favourable conditions, this also makes them difficult to eradicateand manage.
 Cyanobacteria can exist in fresh or marine water, soil or dust and also in many extreme temperatures and habitats. They are found in abundance at the Earth’s frozen polar ice caps and even in the hollow shafts of polar bear’s fur as well as in hot volcanic, sulphurous or salt springs and deserts and in polluted and nutrient rich beaches, lakes, rivers, estuaries, reservoirs and waterways and even inside underwater marine geysers. Photo Credit: Steven Pierson
Cyanobacteria can exist in fresh or marine water, soil or dust and also in many extreme temperatures and habitats. They are found in abundance at the Earth’s frozen polar ice caps and even in the hollow shafts of polar bear’s fur as well as in hot volcanic, sulphurous or salt springs and deserts and in polluted and nutrient rich beaches, lakes, rivers, estuaries, reservoirs and waterways and even inside underwater marine geysers. Photo Credit: Steven Pierson
Cyanobacteria often exist in symbiotic relationships with other life forms such as cycads and fungi and provide valuable food and nutrients for many other organisms but although they are an important part of the biosphere and life cycle they can be a serious nuisance and pest when they form harmful blooms or cyanoHABs and release cyanotoxins that bioaccumulate or poison other organisms and habitats.
The causes of cyanobacterial bloom formation are multifactorial and species specific but they usually include the presence of cells or spores for recruitment, warmer temperatures, high light intensity, low turbidity and stable water and air conditions. When these conditions are present, often but not always in nutrient rich environments known as ‘eutrophication’ cyanobacteria can grow quickly and exponentially to create blooms. Globally, strong correlations have been found between total phosphorous inputs and phytoplankton production in fresh water, and between total nitrogen input and phytoplankton production in estuarine and marine waters. There are also numerous examples of increases in nutrient loading being directly linked with the development of large biomass blooms, leading to anoxia and even toxic or harmful impacts on fisheries resources, ecosystems, and human health through recreational and drinking water.
The connection between eutrophication and an increase in the occurrence of cyanoHABs is well established. Climate changes such as global warming and corresponding hydrological changes have also been implicated (IPCC, 2007, Briand et al., 2004,Elliott et al., 2005, Paerl and Huisman, 2008, Paul 2008, Paerl etal., 2011; cited by Paerl et al., 2012) see figure 1.

figure1 Anthropogenic and ClimateChange Causes of CyanoHaBs (taken from Paerl&Paul, 2012)
Anthropogenic activities and climate change can increase the likelihood of cyanoHAB events occurring but the factors that cause cyanoHABs are manifold and not always fully understood and cyanoHABs can also occur naturally. Modern cyanoHABs are often caused by stronger storms and weather events, increased temperatures and water evaporation rates, reduced water availability and flow, longer droughts and water residence times, increased agricultural, industrial, residential and urban pollution and environmental degradation and waterway eutrophication.
The main sources of nutrients that stimulate algal blooms are sewage, atmospheric deposition, groundwater flow, as well as agricultural and aquaculture runoff. Warming is also a major factor because prokaryotes’ growth rates proliferate at relatively high temperatures (Butterwick et al., 2005 and Watkinson et al., 2005; cited by Paerl&Paul 2012). This also gives CyanoHABs a distinct advantage in nutrient-enriched conditions, when competition with eukaryotic primary producers such as diatoms, chlorophytes, cryptophytes and dinoflagellates is often intense. The growth rates of these eukaryotes usually level off or decline when cyanobacterial growth rates reach their optimum level and then continue to remain high, even when temperatures exceed 25 °C. Surface water warming also intensifies vertical stratification in both freshwater and marine systems and seasonal warming lengthens this period of stratification and cyanoHAB species are able to uniquely exploit these stratified conditions. When blue green algae blooms and cyanoHABs are exploiting lower water levels within the vertical stratification they may no longer be visible on the water's surface but may still be blooming and producing cyanotoxins which are going undetected.
CyanoHABs lower water turbidity in an ecosystem which then restricts light penetration and suppresses the establishment and growth of aquatic macrophytes and benthic microalgae and adversely affects the underwater habitat of benthic flora and fauna (Jeppesen et al., 2007, Scheffer et al.,1997 and Scheffer, 2004; cited by Paerl&Paul 2012). CyanoHAB’s nightime respiration also causes oxygen depletion and bacterial decomposition of dense blooms, which can cause fish kills, and loss of benthic infauna and flora (Paerl, 2004, Watkinson et al., 2005, Garcia andJohnstone, 2006 and Paerl and Fulton, 2006; cited by Paerl&Paul, 2012). Persistent CyanoHABs can lead to long-term loss of benthic habitat (Karlson et al.,2002; cited by Paerl&Paul 2012).
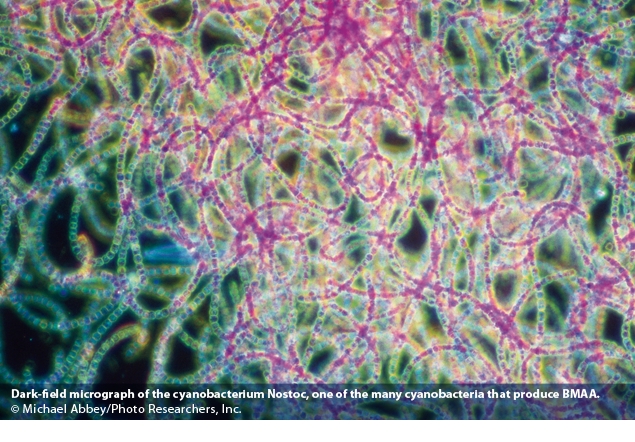
Finally and probably most importantly, cyanoHABs produce toxic peptides and alkaloids and ingestion of cyanotoxins has been linked to liver, digestive and skin diseases, cancers, neurological impairment and neurodegenerative diseases such as Alzheimer’s, Parkinsons and Motor Neuron Disease (MND) ALS or Lou Gehrig's in humans (Holtcamp, 2012, Dunlop etal., 2013) and death (Chorus and Bartram, 1999, Carmichael, 2001 and Humpage, 2008; cited by Paerl&Paul, 2012).
There are five major groups of cyanotoxins produced by cyanoHABs however over 90% of cyanobacteria recently tested have been found to be able to produce this one neurotoxin β-Methylamino-L-alanine (BMAA) during blooming and lysis or cell death and BMAA has been seriously implicated recently as a potential cause of up to 90% of the neurodegenerative diseases occurring within the ALS-dementia complex (Holtcamp,2012, Dunlop et al., 2013). Consequently, toxic CyanoHABs are a major threat to the use of freshwater ecosystems and reservoirs for drinking water, irrigation, and freshwater and marine fishing and recreational purposes (Chorusand Bartram, 1999, Carmichael, 2001, Osborne et al., 2001 and Osborne et al., 2007; cited by Paerl and Paul 2012).
Case Study 1 - LakeAinsworth
Lake Ainsworth is an acidic coastal freshwater lowland dune lake in Lennox Head on the far north coast of NSW and it covers an area of 12.4 hectares, it is classified as ‘permanently’ freshwater, although diatom evidence indicates a saline phase that ended in the 1930s (Tibby 2007, Anon 2004). It is jointly managed by Ballina Shire Council (BSC) and the Department of Sport and Recreation and considered to be an important recreational water asset by BSC and the local community as it provides a safe, closed body of water less than 100 metres from a surfing beach where sports and recreational activities can be undertaken. It is also a significant tourist attraction and brings income and employment to the area. In recent years the lake has been experiencing regular harmful cyanobacterial blooms primarily during the summer months from September to March and this is a threat to public health and safety (Betts 1999, Anon 2002).

Figure 2 taken from the BSC Lake Ainsworth Vegetation Study.
An integrated project report (Jones 1988) of the recreational carrying capacity of Lake Ainsworth with regard to both social and resource capacity identified runoff from the caravan park and surrounding developed areas and alteration of drainage patterns around the lake as potential problems for the future carrying capacity of the lake (Betts 1999, Anon 2002). Jones (1988) also identified beach erosion, leaching from septic tanks and small scale chemical pollution from insect repellant and sun tan lotions and urine from bathers as potential risks to the lake’s water quality in the future, however Jones made no mention of cyanobacteria or cyanoHABs occurring at lake Ainsworth and the water quality was described as ‘pleasant’ ‘tea colour’ and of good quality (Jones 1988,pp.8-24). Since then the water quality has deteriorated significantly and seasonal monitoring has shown that cyanoHABs now occur most years at Lake Ainsworth since the early 1990s (Betts 1999, Anon 2002). Several studies have been performed and the main environmental factors that have been identified as potentially contributing towards the cyanoHABs at lake Ainsworth include light and nutrient availability, water temperature and lake mixing processes (Betts 1999, Anon 2002).
The Ballina Shire Council 2002 Lake Ainsworth Management Report clearly states that ‘better management is needed not only to halt ongoing degradation, but also to rectify past damage,with the overall objective of achieving an integrated, balanced, responsibleand ecologically sustainable use of the area in the future’ (Anon, 2002). To do this the BSC management report (Anon 2002) recommends ‘the establishment of an integrated catchment management structure for the lake that allows for implementation, monitoring and review of the lake Ainsworth Management Plan’ however no subsequent versions of this management plan appear to have been created and the recommended management structure could not be located.
A Lake Ainsworth Monitoring study between 1998-1999 monitored the cyanobacterial environmental conditions and their growth and physiological characteristics and identified the lake stratification in summer months, dune erosion, increased nutrients and faecal contamination as major causes of the prolonged cyanoHAB annual events (Betts, 1999). Betts (1999) also assessed the effectiveness of a 1997 installation of an aerator to improve lake water circulation and decrease stratification and found that it was having some effect (Betts 1999) but cyanoHABs were still occurring for most years (Anon, 2002). BSC recommended more research following a processes study by Australian Water and Coastal Studies in August 1996 (Betts 1999) and at that time the BSC was following the processes outlined in the Estuary Management Manual and the subsequent formation of the lake Ainsworth Estuary Management Committee (Betts 1999).



Figure 3 Figure 4 Figure5
The main groups that are affected by cyanoHABs at Lake Ainsworth are the sports and recreational users of the lake (Anon 2002). This includes tourists, adjacent caravaners, school children at the sports and recreation camp, local bathers, swimmers, fishers, canoeists, catamaran sailors and kayakers who are all advised by BSC signage not to enter the toxic water during prolonged cyanoHAB events mostly during peak summer vacation periods (Betts 1999, Anon 2002).
Although the BSC signs warn of health dangers (see figure 3) there are no fines applying for ignoring this advice and many people and children still enter the water at considerable personal risk, see figures 4 and 5 (Betts 1999, Anon 2002). It has been found recently that over 90% of cyanobacteria and CyanoHABs release the neurotoxin BMAA while they are blooming and dying (Holtcamp, 2012) and that this toxin has been seriously implicated in causing long term neurodegenerative diseases by bioaccumulating in seafood, animals and people and potentially causing the misfolding of important proteins required for healthy neurological functioning (Dunlop et al., 2013). Local authorities say that they have never tested and are currently still not testing Lake Ainsworth’s recreational water for the presence of the neurotoxin Beta-Methylamino-L-alanine or BMAA (BSC, 2014). Lake Ainsworth sports and recreational users who have ignored BSC signage may therefore subsequently develop serious neurodegenerative diseases such as Alzheimer’s, Parkinsons and Motor Neuron Disease and/or liver diseases and cancers, months or years after accidentally ingesting the invisible BMAA and other cyanotoxins present in the lake water during and after the harmful blue green algal blooms or cyanoHABs (Holtcamp, 2012).
CaseStudy 2 – Emigrant Creek Dam
At the time of writing this (May 2014) Emigrant Creek Dam has a blue green algae red alert in place from the Northern Rivers Regional Algae Coordinating Committee (NSW RACC 2014). Emigrant Creek Dam (ECD) is a secondary source of potable and drinking water supplied by Rous Water (Akhurst, 2013) that is located close to Ballina (see figure 2). ECD has a surface area of 0.5 km2, a maximum depth of 9.5 m and a storage capacity of 820 ML (Akhurst, 2013).
Emigrant Creek Dam was built in 1968 and supplies drinking water to the local authority areas of Ballina and Lennox Head (see figure 5) and is an important part of the Rous Water supply Network (Akhurst, 2013). The catchment area for Emigrant Creek Dam contains pasture, macadamia plantations, native bush land, orchards, rural residential and cropping (SKM,2004; cited by Akhurst,2013). The surface water quality in the ECD has deteriorated significantly over recent years (Egis, 2001; SKM, 2004; cited by Akhurst, 2013) and the eutrophic ECD reservoir receives high quantities of nutrient rich water from Emigrant Creek arising from these agricultural holdings. In addition, many small tributaries, land-disposed dredged sediment, a stockpile of harvested aquatic macrophytes on the north-western shoreline and a disused cattle dip site all provide point sources that pollute the water quality andupset the biodiversity of the Emigrant Creek Dam (Akhurst, 2013).
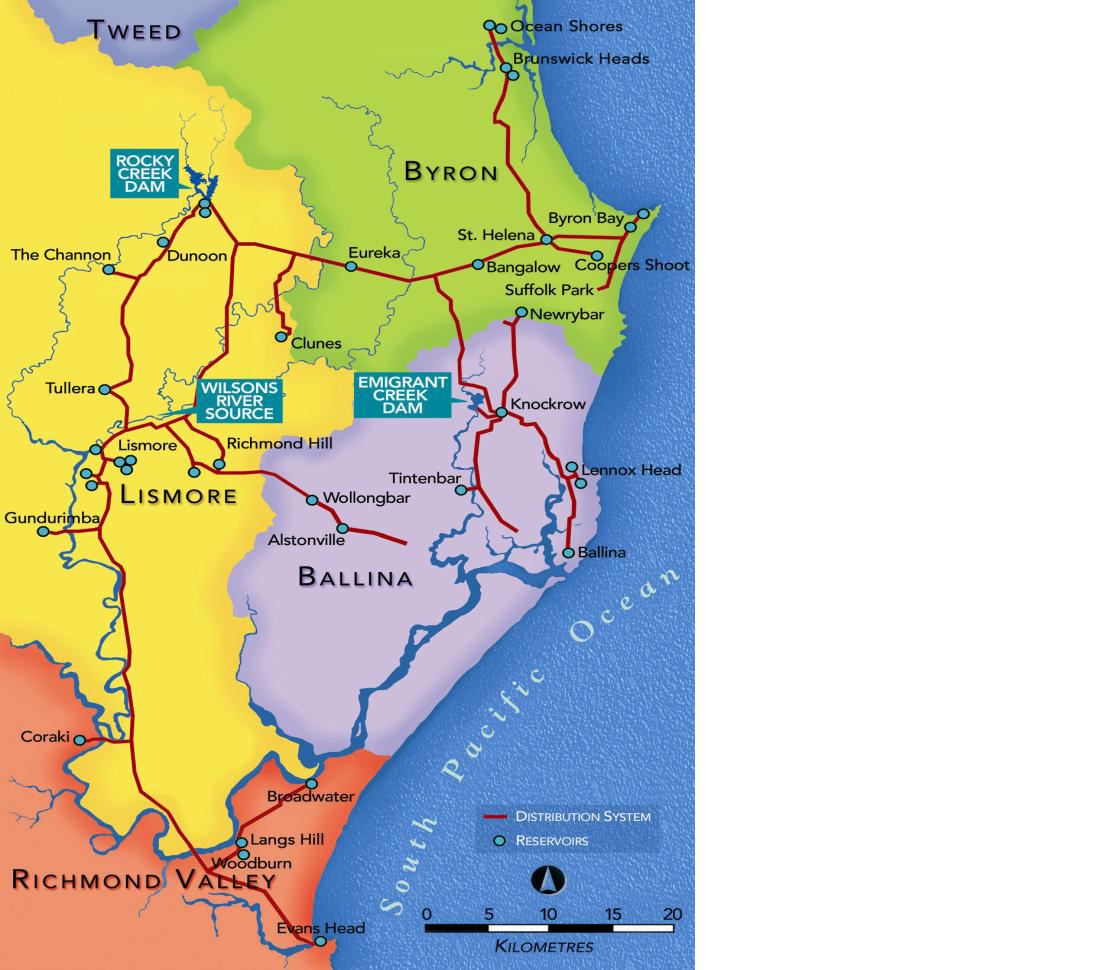
Figure6 © Rous County Council 2011.
Frequent blooms of nuisance cyanobacteria, high exotic aquatic macrophytes biomasses, low dissolved oxygen and elevated concentrations of nutrients and trace metals, including Al, Cu, Fe and Mn originating from seasonal internal elemental ECD recycling of sediments, are all major problems at Emigrant Creek Dam and are making management and treatment of the drinking water difficult (Egis, 2001; Akhurst,2013). Nutrient and trace metal loads in ECD sediments and the overlying water column are directly linked to reservoir eutrophication and a proliferation of potentially toxic cyanobacteria and their cyanoHABs. High accumulations of trace metals and/or nutrients may result in serious ecological degradations (Saygi and Yiğit, 2012; cited by Akhurst, 2013).
Clean drinking water resources are vital for all modern societies; however the presence of potential pollutants and cyanoHABs in the Emigrant Creek Dam are a potential health risk not only for aquatic environment degradation but also for animal and human health (Akhurst, 2013). A drinking water quality risk assessment (Egis,2001; cited by Akhurst, 2013) and a water monitoring program in Emigrant Creek(SKM, 2004; cited by Akhurst, 2013) and an aquatic and terrestrial biota survey (EEP, 2003; cited by Akhurst, 2013) were all performed for the ECD; however no reservoir-wide assessments of the ecological risks associated with sediment accumulated elements have been made (Akhurst, 2013). There are also no tests being performed by Rous Water or the NGO’s that they employ for the presence of the cyanotoxin BMAA in the ECD water which can still remain after cyanobacteria blooms have died or been removed by activated carbon and ozone treatments (Rous Water, 2014; Richmond QLD Forensic Department, 2014; House et al., 2010). Blue green algae was recently detected in the Emigrant Creek at Tintenbar below the ECD and this creek empties into the Richmond River in Ballina where commercial and recreational fishing and bathing also takes place and where the largest fish kill in Australia occurred in 2001 and another in 2008. 'Blackwater' and resultant anoxia after floods were blamed along with acid sulphate run-off however the latter was later dismissed as not being the cause. No records of any tests done during this period for the presence of blue green algae blooms, cyanobacterial cell death or lysis or neurotoxic BMAA could be found.
Policies
Information and advice regarding the existence and management of cyanoHABs in Australian recreation and drinking water is potentially available to the public and to those who are responsible for managing the problem from a variety of global, national, state and local governmental and non-governmental organizations (NGOs) ranging from international, national and state government departments, catchment authorities and NGOs to local government, councils and the NGOs that they employ (Verhoeven, 2014). However, the names and hierarchical structure of these organizations and the advice and information that they provide are constantly being revised and changed as is the latest scientific research and advice so it is difficult to outline clearly who is responsible for managing cyanoHABs in recreation and drinking water at any one point in time, especially as links and references to relevant advisory sites and documents are also constantly changing with each successive government departmental restructure, merge or change in hierarchy and those references and links are often not being maintained.
Examples of global organizations that research and provide global advice on managing cyanoHABs in recreational and drinking water include UNESCO, WHO, GWRA and the EPA (Newcombe et al., 2009). The Australian national organization that primarily researches and provides national advice on managing cyanobacteria and cyanoHABs in recreational and drinking water is Water Quality Research Australia (WQRA formerly WATERRA), which is based in South Australia (Newcombe et al., 2009). Ultimately the responsibility for cyanoHABs occurring in recreational water and drinking water and the consequences of potential mismanagement seems to lie primarily with local governments, catchment authorities and county councils and the water utilities and consultants that they employ, who rely upon various global, national, state and local governmental and NGO water organizations and experts to supply them with safe recreational and drinking water advice and guidelines regarding cyanobacterial and cyanoHAB management, treatment and monitoring services.
The local authorities ability to manage cyanoHABs in their recreational and drinking water appears to be primarily self reported and self-audited although each of the Australian states and territories do have their own hierarchical infrastructure of governmental departments and authorities and NGOs responsible for monitoring the local and regional management of cyanobacteria and cyanoHABs. For example in NSW the Government Department of Primary Industry oversees and manages various regional algal coordinating committees, catchment authorities and councils that in turn research and monitor cyanoHAB events and are responsible for reporting these cyanoHAB events and their level of harm often as a colour coded level of alert in recreational and drinking water to the public, state and national authorities (NSW RACC 2014).
The National Water Commission (NWC) that was responsible for auditing the state of Australian recreational and drinking water quality was recently abolished by the current Australian federal government as part of its financial ‘budget’ and cost saving policies, saving $20.9 million (Murray, 2014). The NWC was an independent statutory authority established by the National Water Commission Act 2004 to provide advice to the Council of Australian Governments (COAG) and to the Australian Federal Government on national water issues (National WaterCommission (NWC), 2011). It was responsible for providing independent assurance of governments' progress on water reform and promoting the objectives and outcomes of the National Water Initiative, which was an agreement signed by all governments in Australia (NWC, 2011). Since a COAG review in 2012 and the amendment of the Nation Water Commission Act the NWC was responsible for monitoring, auditing and assessing the state of the Nation’s water resources and their management and was empowered to perform broader activities to promote national water reform objectives (National Water Commission, 2013). The NWC also had additional functions under other Commonwealth acts and regulations, for example the Water Act 2007 authorised the NWC to audit the effectiveness of the implementation of the Murray–Darling Basin Plan and associated water resource plans (NWC, 2011). In 2011, the NWC was also delegated additional functions under the Carbon Credits (Carbon Farming Initiative) Regulations 2011 (NWC, 2013).
Diffuse source water is a major contributor to nutrient pollution, eutrophication and cyanoHAB events in Australian waterways (NSW DSWP, 2011). The NSW Diffuse Source Water Pollution Strategy (NSW DSWP, 2011) has provided, for the first time, a management framework for coordinating efforts to reduce diffuse source water pollution across NSW. It is a joint initiative by the State's natural resource managers at State, regional and local government levels that promotes partnerships and provides a means to share information on projects and their outcomes across the State (NSW DSWP, 2011). The main aim of the Strategy was to reduce diffuse source water pollution inputs into all NSW surface and groundwater and to contribute towards the community agreed NSW water quality objectives and state-wide natural resource management targets to progress towards Goal 22 (Protect our natural environment) of ‘NSW 2021 -A plan to make NSW number one’ (NSW DSWP, 2011).
The first and also the last NSW Diffuse Source Water Pollution Strategy Annual Report was published in November 2010 and reported on the implementation of the individual actions identified it the Priority Action Plan. It claimed that very few projects had been delayed in their implementation and completion and a range of newinitiatives and that projects had been identified that will continue to buildon the substantial achievements to date (NSW DSWP, 2011).
Conclusion
The case studies in this paper have shown how in just one small northern NSW Australian town such as Ballina, cyanobacteria and dangerous cyanoHABs are occurring regularly and their management is proving to be very challenging to local, state and federal authorities and to the NGOs that they employ. Known cyanotoxins from cyanoHABs such as BMAA that have recently been shown to be potentially causing up to 90% of neurodegenerative diseases such as Alzheimer’s, Parkinsons and Motor Neuron disease are possibly occurring in many of our local recreational and drinking water supplies and yet their presence is reportedly not being tested for by the relevant authorities. The Australian Bureau of Statistics (ABS) do not currently have health statistics to show how many people are dying from these neurodegenerative diseases in specific locations potentially because of their direct exposure to cyanoHABs and cyanotoxins such as BMAA in their recreational and drinking water supplies. More research is needed and better monitoring and auditing of recreational and drinking water cyanoHABs. The abolishment of the National Water Commission by the federal government this year may well prove to have been a false economy for the Australian public.
The evidence in this paper supports Dr Paul Cox and his colleagues recommendations that people and governments should take the threat of BMAA neurotoxins and other cyanotoxins from cyanobacteria and cyanoHABs in our recreational and drinking water supplies much more seriously (Holtcamp, 2012). Federal, state and local governments, catchment authorities, the public and NGO water managers and consumers need to take a closer look at cyanobacterial blooms and the environmental and health effects of cyanoHABs and in our recreational and drinking water that may be bioaccumulating in our food and ultimately in us (Holtcamp, 2012).
“Australia needs to have a vigorous public discourse as to how we manage our land and water and scientists and academics, funded from the public purse, have an obligation to contribute to this discourse. All Australians must become more water literate in order to understand the impact they have on our water resources” (Cullen, 2011).
References
Akhurst, D. J. (2013). Sediment geochemistry and short-term biomanipulations in Emigrant Creek Dam, New South Wales, Australia.
Anon.(2002) Lake Ainsworth management plan. Ballina (NSW), Department of Public Works and Services, Geolink, Ballina Shire Council, NSW, Australia. http://www.ballina.nsw.gov.au/cp_content/resources/12_30443__Lake_Ainsworth_Management_Plan.pdf(Viewed May 2014)
Anon.(2004) Unsolved mystery: clues wanted on Lake Ainsworth’s salty past. NorthCoast Advocate 22 Jan 2004: 1–2.
Betts, J. (1999). Lake Ainsworth monitoring study : final report. Ballina Shire Council. NSW Department of Public Works and Services, Geolink.
Cullen, P.,&Cullen, V. (2011). This Land, Our Water: Water Challenges for the 21st Century. ATF. http://www.nrc.nsw.gov.au/content/documents/Speaking%20notes%20-%20This%20Land%20Our%20Water.pdf (viewed May 2014).
Dunlop, R.A., Cox, P.A., Banack, S.A., Rodgers,J.K. (2013). "The Non-Protein Amino Acid BMAA Is Misincorporated into Human Proteins in Place of l-Serine Causing Protein Misfolding and Aggregation". PLOS One 8 (9). doi:10.1371/journal.pone.0075376.
Holtcamp, W. (2012). The emerging science ofBMAA: do cyanobacteria contribute to neurodegenerative disease?. Environmental health perspectives,120(3), a110.
House, J., Ho, L., Baker, P.,&Burch, M.(2010). Management strategies for cyanobacteria (blue-green algae): A guide for water utilities.WQRA Research report 74 [www document]. http://www.awa.asn.au/uploadedfiles/Content/Training_and_Development/Online_Training/Leaks,Smells%20and%20Corrosion/Management_Stratetgies_for_Cyanobacteria_Guide_for_Water_Utilities.pdf(Viewed May 2014)
Murray D. (2014) ‘What does the first Abbott government budget mean for our environment?’ Australian Conservation Society[web address] http://www.acfonline.org.au/news-media/acf-opinion/what-does-first-abbott-government-budget-mean-our-environment
Newcombe G., House J., Ho L., Baker P. and Burch M. (2009) Management Strategies for Cyanobacteria (Blue‐GreenAlgae) and their Toxins: A Guide for Water Utilities. WQRA research report 74. http://www.wqra.com.au/WQRA_publications.htm (ViewedMay 2014)
Newcombe G., (2011) International GuidanceManual for the Management of Toxic Cyanobacteria (SA Water Corporation, WaterQuality research Australia WATERRA&Global water Research Coalition GWRC).ISBN 978–90–77622–21–6 http://www.waterra.com.au/cyanobacteria-manual/PDF/GWRCGuidanceManualLevel1.pdf(Viewed May 2014)
National Water Commission NWC. (2011) The National Water Initiative-securing Australia‟ s water future: 2011 assessment. Canberra,NWC.
National Water Commission. (2013). The NationalWater Commission Role and Functions. Canberra, NWC. http://www.nwc.gov.au/organisation/role(Viewed May 2014).
NSW DSWP Government Department of Environmentand Heritage (DEH) (2011) NSW Diffuse Source Water Pollution (DSWP) Strategy http://www.environment.nsw.gov.au/water/dswp.htm(Viewed May 2014)
NSW RACC (2104) Department of Primary Industries Regional Algal Coordinating Committee Contacts http://www.water.nsw.gov.au/Water-management/Water-quality/Algal-information/Algal-contacts/Algal-contacts(Viewed May 2014)
Rous Water Management Plan (Anon 2014) URL www.rouswater.nsw.gov.au (ViewedMay 2014)
Tibby, J., Lane, M. B.,&Gell, P. A.(2007). Local knowledge and environmental management: a cautionary tale fromLake Ainsworth, New South Wales, Australia. EnvironmentalConservation, 34(04),334-341.
Verhoeven, TJ. New South Wales Algal Management Strategy : Integrating the Professions and Community [online]. In: Water Down Under 94: Groundwater/Surface Hydrology Common Interest Papers; Preprintsof Papers. Barton, ACT: Institution of Engineers, Australia, 1994: 73-77.National conference publication (Institution of Engineers, Australia) ; no.94/10.Availability: <http://search.informit.com.au/documentSummary;dn=747610112540262;res=IELENG>ISBN:085825607X. (Viewed May 2014)
Whitton, B. A.,&Potts, M. (Eds.).(2000). The ecology of cyanobacteria: their diversity in time and space. Springer.




