There are a number of things which can damage DNA - toxic chemicals are the primary concern on earth but, in space, ionized radiation is the worry. Unrepaired DNA can lead to mutations, which in turn can lead to diseases like cancer. Intricate DNA repair mechanisms in the cells' nuclei are constantly working to fix what's broken, but whether the repair work happens "on the road" — right where the damage occurs — or "in the shop" — at specific regions of the nucleus — is an unanswered question.
NASA scientists are working on understanding where and how DNA is repaired so they can be prepared for long distance space travel. By comparing computer models of damaged human DNA with microscopic images of human cells that reveal focal sites of radiation-induced damage, researchers in the Life Sciences Division (LSD) of the Lawrence Berkeley National Laboratory, with colleagues at NASA and the Universities Space Research Association, have found evidence that there are specific regions where broken DNA is concentrated for repair.
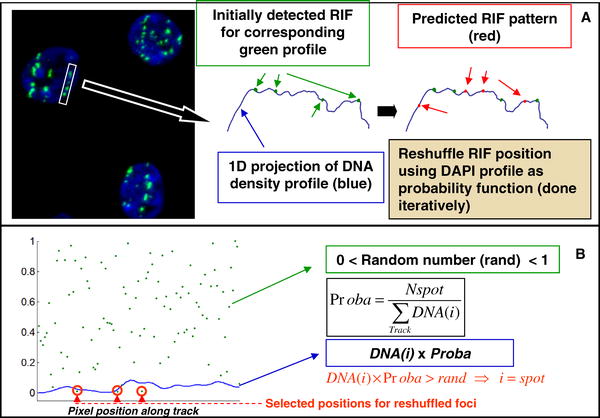
(A) Shows a typical image of cells that have been traversed with 1 Gy of 1 GeV/amu Fe ions. After manually selecting a region that contains a clear track, foci identification and reshuffling is done as depicted by the cartoon. Foci detection is done automatically via in-house image algorithm (see Materials and Methods).
(B) Further illustrates the mathematical approach used (i.e., Monte Carlo concept), where the probability of damage at a pixel location is proportional to the DNA density at the same location. This process is done iteratively (i.e., 50 randomizations per nucleus analyzed) to give a reasonable average break distribution. For each iteration, RIF position is determined by a probability less than that determined by DNA density (blue line).
"NASA has long been interested in the radiation hazards in space," says LSD's Sylvain Costes, who led the study. "On a trip to Mars, astronauts will be exposed to cosmic rays for as long as three years, so NASA has been trying to come up with a mechanistic model of DNA repair to estimate the increased risk of cancer. We are helping to develop such a model."
Double-strand breaks and radiation-induced foci
In many NASA studies cells have been exposed to particles like those found in cosmic rays, such as energetic iron nuclei produced in accelerators at Brookhaven National Laboratory. The goal is to determine how many double-strand breaks (DSBs) — in which both strands of the DNA double helix are severed — occur per gray of radiation. (One gray is equivalent to 100 rads, an older unit signifying "radiation absorbed dose"; a gray equals one joule of energy absorbed per kilogram of matter.)
DSB yield can be measured by pulling apart a cell's radiation-severed DNA using gel electrophoresis: the shorter the fragments in the gel bands, the more frequent the breaks. Interpreting the band patterns thus leads to an estimate of the average number of breaks per cell for a given dose and time following exposure. NASA has developed a computer model based on these measured DSB yields to predict DSB formation in hypothetical human cell nuclei.
On the other hand, gel electrophoresis fails to indicate the severity of the damage, or where in the nucleus the double-strand breaks occur. The default assumption has been that DSBs occur randomly in a homogenous distribution of DNA in the nucleus.
Most gel electrophoresis studies indicate about 25 to 30 DSBs per gray of gamma rays. In microscopic images of real cells, however, the visible sites that might be assumed to correspond to double-strand breaks — sites called "radiation induced foci," or RIF — occur at a lower rate, only about 15 per gray, depending on the cell type.
"What we see through the microscope is not the broken DNA itself but a collection of proteins associated with breaks, which we have labeled with fluorescent stains. These include modified histones, which are part of the chromosomal material, and other proteins that seek out DNA breaks and recruit repair machinery to fix them," says Costes. "The first question we had to answer was how closely RIF are associated with DSBs."
Costes recognized that one way to test the association was to calculate how energy was deposited by different kinds of radiation. A high-energy particle, for example, moves through the nucleus in a straight line and may strike DNA at any point along the way; the pattern of hits along the track should be essentially random.
"Physicists understand very well how energy is deposited in the cell's DNA," says Costes, "but the challenge was to make a predictive model that was consistent with what a biologist sees through the microscope. To test what should be seen, given a specific radiation dose, I set out to turn NASA's high-resolution DNA damage simulator" — a model based on data from gel electrophoresis — "into artificial microscope images of a nucleus damaged by radiation."
To do this, says Costes, "I simulated the optical transformation when imaging events at the nanoscale are viewed with a fluorescent microscope." One optical transformation is blurring, with the result that, in a real microscope, two nearby radiation-induced foci may blend together and look like one.
When such factors were taken into account, Costes's initial model, following the NASA model, confirmed random distribution of double-strand breaks at the micron scale. The frequency of DSBs was radiation dependent, however: in the case of cosmic rays (NASA's main concern), which lead to complex double-strand breaks, there was good agreement with the RIF frequencies seen in the microscope. But in the case of gamma rays, which lead primarily to sparse and noncomplex DSBs, measured frequencies in real microscope images were lower.
This suggests that RIF correspond to sites of complex double-strand breaks in DNA. Moreover — focusing on cosmic rays for the NASA project — although the DSB frequency was the same as predicted by the models, the RIF in real microscope images were distributed very differently.
Model physics, real biology
"For one thing, there is a time effect," says Costes. "Just five minutes after cells are exposed to high-energy particles, microscope images already show a nonrandom distribution of RIF." The RIF occur along straight lines, as expected for a particle track, but are not randomly distributed along the lines. "Even though we have the right foci frequency along a track, many foci appear to repulse each other within the first 30 minutes after irradiation" — a suggestion that they might be moving to specific regions of the nucleus.
Costes and his colleagues applied the same kinds of models and measurements to gamma rays and found that whereas the model predicted that these breaks would occur in a random pattern in three dimensions throughout the nucleus (not along lines), in fact, under the microscope, radiation-induced foci caused by gamma rays were also distributed nonrandomly. There were fewer RIF due to gamma rays than the model predicted, and they appeared more gradually.
The researchers now used a novel technique, "relative DNA image measurements," and analyzed the images to show where the DNA was dense, where it was less dense, and where the two densities met. They wanted to see whether, where RIF occurred, there was an underlying difference in the nature of the DNA.
Indeed there was. By comparison to the random distribution that might have been expected, RIF were more frequent in the low-density regions. And a preponderance of RIF coincided with the interfaces between the two kinds of DNA.
"In terms of physics, there's no obvious explanation for this observation," says Costes. "In terms of biology, we have some pretty good guesses."
What's evident is that the organization of the cell nucleus plays an important role in response to DNA damage. The high-density regions likely are heterochromatin, the parts of the chromosomes where genes are (for the most part) silenced. The low-density regions likely correspond to euchromatin, the parts of the chromosomes where most genes are actively transcribed. While it's possible that damage in condensed regions causes the condensed chromatin to open up and appear less dense, it is more likely, Costes believes, that the damaged sections actually migrate toward the less-dense chromatin and concentrate at the high-density/low-density interface.
Researchers have previously proposed the existence of "repairosomes" in mammalian cells, similar to specific regions where DNA is repaired in yeast, although these have never been observed in mammals. The fact that RIF concentrate in specific regions of the human cell nucleus — and apparently tend to move toward shared sites — is highly suggestive of such repair centers, where activities like the necessary clamping and orientation of the broken strands take place.
"Our results raise a number of interesting possibilities," says Costes. "If less-dense regions of the nucleus are more susceptible to radiation damage, we may be able to use this knowledge to make tumor cells easier to kill. Repair centers in mammalian cells are probably an efficient way to repair the typically sparse and noncomplex DNA damages of daily cellular life."
But there's a downside, Costes says. "Humans didn't evolve with cosmic rays, and the existence of repair centers may not be good news for astronauts — cosmic rays primarily generate spatial clusters of complex DNA breaks. If these breaks are gathered in common locations in the nucleus, they will most likely lead to chromosomal translocation, a process thought to play an initiative role in cancer."
Source: Image-based modeling reveals dynamic redistribution of DNA damage into nuclear sub-domains, by Sylvain V. Costes, Artem Ponomarev, James L. Chen, David Nguyen, Francis A. Cucinotta, and Mary-Helen Barcellos-Hoff






Comments