A study in Science has tied the aging process to the deterioration of tightly packaged bundles of cellular DNA known as heterochromatinand say that the genetic mutations underlying Werner syndrome, a disorder that leads to premature aging and death, results in the deterioration of these bundles.
The discovery used stem cell and gene-editing technologies and could lead to ways of countering age-related physiological declines by preventing or reversing damage to heterochromatin.
Werner syndrome is a genetic disorder that causes people to age more rapidly than normal. It affects around one in every 200,000 people in the United States. People with the disorder suffer age-related diseases early in life, including cataracts, type 2 diabetes, hardening of the arteries, osteoporosis and cancer, and most die in their late 40s or early 50s. The disease is caused by a mutation to the Werner syndrome RecQ helicase-like gene, known as the WRN gene for short, which generates the WRN protein.
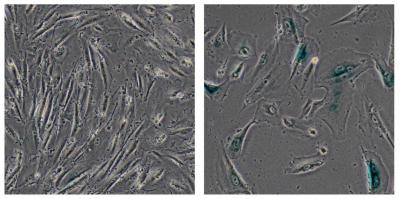
This image shows normal human cells (left) and genetically modified cells developed by the Salk scientists to simulate Werner syndrome (right), which showed signs of aging, including their larger size. Credit: Salk Institute
Previous studies showed that the normal form of the protein is an enzyme that maintains the structure and integrity of a person's DNA. When the protein is mutated in Werner syndrome it disrupts the replication and repair of DNA and the expression of genes, which was thought to cause premature aging. However, it was unclear exactly how the mutated WRN protein disrupted these critical cellular processes.
In the study, the researchers sought to determine precisely how the mutated WRN protein causes so much cellular mayhem. To do this, they created a cellular model of Werner syndrome by using a cutting-edge gene-editing technology to delete WRN gene in human stem cells. This stem cell model of the disease gave the scientists the unprecedented ability to study rapidly aging cells in the laboratory.
The resulting cells mimicked the genetic mutation seen in actual Werner syndrome patients, so the cells began to age more rapidly than normal. On closer examination, the scientists found that the deletion of the WRN gene also led to disruptions to the structure of heterochromatin, the tightly packed DNA found in a cell's nucleus.
This bundling of DNA acts as a switchboard for controlling genes' activity and directs a cell's complex molecular machinery. On the outside of the heterochromatin bundles are chemical markers, known as epigenetic tags, which control the structure of the heterochromatin. For instance, alterations to these chemical switches can change the architecture of the heterochromatin, causing genes to be expressed or silenced.
Salk professor Juan Carlos Izpisua Belmonte explains how his lab tied the aging process to the deterioration of tightly packaged bundles of cellular DNA. This new work could lead to methods of preventing and treating age-related diseases such as cancer, diabetes and Alzheimer's disease. Credit: Salk Institute
"Our findings show that the gene mutation that causes Werner syndrome results in the disorganization of heterochromatin, and that this disruption of normal DNA packaging is a key driver of aging," says Juan Carlos Izpisua Belmonte of the Salk Institute, a senior author on the paper. "This has implications beyond Werner syndrome, as it identifies a central mechanism of aging--heterochromatin disorganization--which has been shown to be reversible."
The researchers discovered that deletion of the WRN gene leads to heterochromatin disorganization, pointing to an important role for the WRN protein in maintaining heterochromatin. And, indeed, in further experiments, they showed that the protein interacts directly with molecular structures known to stabilize heterochromatin--revealing a kind of smoking gun that, for the first time, directly links mutated WRN protein to heterochromatin destabilization.
"Our study connects the dots between Werner syndrome and heterochromatin disorganization, outlining a molecular mechanism by which a genetic mutation leads to a general disruption of cellular processes by disrupting epigenetic regulation," says Izpisua Belmonte. "More broadly, it suggests that accumulated alterations in the structure of heterochromatin may be a major underlying cause of cellular aging. This begs the question of whether we can reverse these alterations--like remodeling an old house or car--to prevent, or even reverse, age-related declines and diseases."
More extensive studies will be needed to fully understand the role of heterochromatin disorganization in aging, including how it interacts with other cellular processes implicated in aging, such as shortening of the end of chromosomes, known as telomeres.
Other authors on the paper include: Weiqi Zhang, Jingyi Li, Keiichiro Suzuki, Jing Qu, Ping Wang, Junzhi Zhou, Xiaomeng Liu, Ruotong Ren, Xiuling Xu, Alejandro Ocampo, Tingting Yuan, Jiping Yang, Ying Li, Liang Shi, Dee Guan, Huize Pan, Shunlei Duan, Zhichao Ding, Mo Li, Fei Yi, Ruijun Bai, Yayu Wang, Chang Chen, Fuquan Yang, Xiaoyu Li, Zimei Wang, Emi Aizawa, April Goebl, Rupa Devi Soligalla, Pradeep Reddy, Concepcion Rodriguez Esteban, Fuchou Tang and Guang-Hui Liu. Funding for the study was provided by the Glenn Foundation, the G. Harold and Leila Y. Mathers Charitable Foundation and the Leona M. and Harry B. Helmsley Charitable Trust.






Comments