Scientists at the U.S. Department of Energy's Lawrence Berkeley National Laboratory (Berkeley Lab) and the University of California at Berkeley have performed the first scanning tunneling spectroscopy of graphene flakes equipped with a "gate" electrode. The result is the latest in a series of surprising insights into the electronic behavior of this unique, two-dimensional crystal form of carbon: an unexpected gap-like feature in the energy spectrum of electrons tunneling into graphene's single layer of atoms.
Michael Crommie, a faculty scientist in Berkeley Lab's Materials Sciences Division and a professor in the Department of Physics at UC Berkeley, explains that this peculiar feature of the electronic structure of graphene arises from the interaction of the tunneling electrons with phonons, the quantized vibrations of the 2-D graphene crystal, and may lead to novel applications for future graphene nanodevices.
A team led by Yuanbo Zhang, a postdoctoral fellow in Crommie's research group, discovered graphene's mysterious energy gap; the research appears in advanced online publication on the Nature Physics website at http://dx.doi.org/10.1038/nphys1022.
"Monolayer graphene – flakes a single atom thick — were first isolated by Andrew Geim's group at the University of Manchester, England, in 2005, and have been intensely studied since then," says Crommie. "Graphene's interesting electronic effects opens a new realm of basic science. It's an entirely new material, with new physics that could lead to new practical devices and applications. In that respect it's as promising as carbon nanotubes — but graphene's planar geometry is potentially even more versatile."
Crommie says, "Because graphene is two-dimensional, it can be carved up and cut into tailored shapes, like cutting a sheet of paper." The shape might include features like narrow sections to control the flow of electrons, edges with unique magnetic properties, and dopant atoms implanted at precise locations in the 2-D matrix.
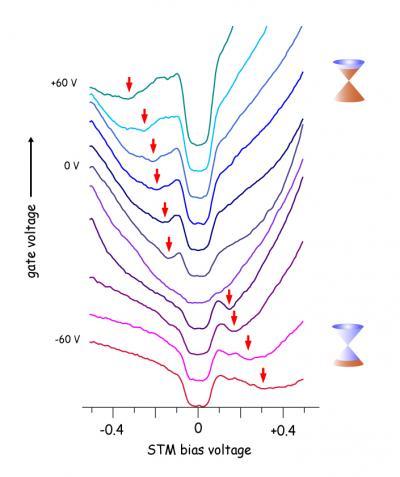
These STM electron tunneling spectra were obtained at the same physical location on a graphene surface held at different gate voltages. The vertical scale gives a measure of the energy-dependent graphene electron density of states; the different curves result from the different applied gate voltages. Changing the gate voltage allows the density of charge carriers in the graphene to be controllably varied. The red arrows indicate the measured tunnel current signal that occurs when electrons tunnel from the STM tip to the graphene Dirac point, a minimum in the graphene density of states.
Photo Credit: Lawrence Berkeley National Laboratory
"Two-dimensionality confers an amazing degree of flexibility," he says, "but to take full advantage of this new material, we will need to understand what is happening at atomic length scales. That's where the STM — the scanning tunneling microscope — comes in."
Studying gated graphene with the STM
The business end of the STM is the tip, a fine metal wire placed in close proximity to a conducting surface — in this case a flake of graphene contacted by thin metal electrodes. An applied voltage between the tip and sample causes electrons to tunnel between them — a "tunnel current." At constant voltage the tunnel current depends on the position of the tip with respect to the surface, so by scanning the tip across the flake the surface topography can be mapped.
The current can also be varied by changing the voltage between tip and surface, which gives information about the electronic structure of the material — in particular the local density of states (LDOS, an energy-dependent electron density) below the tip. Combining STM microscopy and spectroscopy allows a researcher to construct an image of the spatial distribution of the electronic states.
The Crommie group's experiments used exfoliated graphene, individual flakes made by mechanically cleaving a sheet of atoms from a larger chunk of carbon. The group attached electrodes to both the graphene flake and an underlying substrate consisting of a conducting layer of silicon, which was separated from the flake by an insulating layer of silicon dioxide. The experimental setup was thus able to uniquely incorporate two distinct voltage differences, that between the tip of the STM and the surface (the "bias" voltage) and that between the graphene flake and the underlying substrate (the "gate" voltage).
"The purpose of controlling the gate voltage is to vary the density of the charge carriers in the graphene," Crommie says. "The purpose of varying the STM bias voltage is to perform spectroscopy, so we can look at the graphene's local density of states at different energies. We want to know where are the electrons? How are they behaving?"
These questions are of particular interest because of graphene's odd electronic properties. The carbon atoms in graphene are arranged at the corners of hexagons, as in chicken wire, with three of each atom's four electrons involved in molecular bonds with its neighbors; these are sigma orbitals that lie in the plane of the material. The remaining electrons are in pi orbitals extending above and below the plane. The hybridization of the pi orbitals spreads across the graphene sheet, and the unconfined electrons are free to move as high-speed "relativistic quasiparticles," so-called Dirac fermions which act as if they have no mass.
The plot of energy states for Dirac fermions in graphene looks quite different from that of a conventional 3-D semiconductor, which typically consists of two opposing parabolic curves, a lower-energy valence band and a higher-energy conduction band, with a band gap between them that no charge carriers can occupy.

Melissa Panlasigui, Michael Crommie, Yuanbo Zhang, and Victor Brar are among the members of Crommie's group and colleagues who discovered graphene's phonon floodgate. Not pictured are authors Feng Wang, Caglar Girit, Yossi Yayon, and Alex Zettl.
Photo Credit: Roy Kaltschmidt, Lawrence Berkeley National Laboratory
Graphene's unusual electronic properties
By contrast, the Dirac fermion energy states of graphene can be represented as two cones with their vertices meeting at a point of minimum electronic density, called the Dirac point. Thus one might expect the spectrum of the density of states resulting from electrons tunneling into graphene to be linear, following the smooth edge of the touching cones.
"When we plotted the LDOS spectra of our gated graphene flakes, however, we found a gap-like feature that was centered on the Fermi energy — no matter how we changed the density of charge carriers in the graphene with the gate voltage and no matter where we looked on the flake," Crommie says.
The Fermi energy is the energy of the highest occupied electronic state in a graphene flake, and is the reference energy for this kind of measurement. "Almost no electrons tunneling from the STM tip could enter the graphene at low energies within this gap region, but at slightly higher energies there was an abrupt, giant enhancement in tunneling, like a floodgate opening up for electrons." And this was not the only odd feature in the graphene spectrum.
"There was another feature in the spectrum, a local minimum of states, which moved in a very regular way as we changed the gate voltage and thus the density of charge carriers in the material," Crommie says. The research team was able to unambiguously identify this feature as the mark of electrons tunneling from the STM tip to the Dirac point itself, the minimum in graphene density of states.
And what of the mystery gap itself? "We realized that this is not a true energy gap; it is not a feature of the electronic band structure of graphene," Crommie says. "Rather it marks the interaction of the tunneling electrons with phonons, the quantized vibrations of the graphene lattice."
Naturally occurring vibrations in the graphene sample are minimal for the Crommie group's STM setup, since it is kept very cold (just four degrees above absolute zero). However, when the bias voltage between the tip and graphene sample increased above a special threshold of 63 millivolts, "then each tunneling electron is able to create a phonon vibration in the graphene sheet, which allows the electron to get into the graphene much easier," Crommie says.
Indeed, this "phonon-assistance" causes the electron tunneling conductance to suddenly increase by more than 10 times, as phonons essentially open a new channel for electrons to flow through. Says Crommie, "We call it a phonon floodgate."
An underlying cause for this new channel arises from the carbon sigma orbitals, which normally don't conduct electrons (as the pi orbitals do), but which are brought into play when the graphene sheet vibrates. "When a phonon is created, the sigma orbital kind of rubs up against the pi orbital and acts like grease to help insert a tunneling electron into graphene," Crommie says.
"We started this research by simply asking, what do you see when you measure a graphene device with STM?" Crommie says. "In the process, we discovered a completely unexpected phonon floodgate. This gives us new insight into how electrons and phonons behave in graphene and creates new opportunities for future graphene-based nanodevice applications."
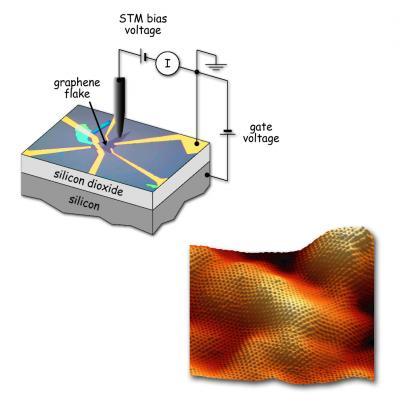
Under the STM tip a flake of graphene 50 microns (millionths of a meter) long rests on a substrate of silicon with a thin layer of silicon dioxide insulation (upper left). The graphene is contacted by gold electrodes, connected to outside experimental equipment and to the underlying silicon electrode, which is used to apply a gate voltage. At lower right, a topographical image of the graphene flake, 10 nanometers (billionths of a meter) across. Variations in height of one or two angstroms (ten-billionths of a meter) are likely caused by the roughness of the substrate.
Photo Credit: Lawrence Berkeley National Laboratory






Comments