The laws of thermodynamics dictate a simple policy of universal application. It is one that policy makers must be forced to understand: mother nature will not permit us to reverse a fossil fuel energy production process without using more energy than the process has itself released.
One of the most ludicrous ideas I have ever read about, purportedly coming from scientists, is that we can somehow sequester carbon. Let me give my reasons.
Coal and oil have varying, but high, carbon content. When they are burned they produce vast quantities of heat energy and CO2. To split the CO2 into carbon and oxygen would require more energy input than was obtained as output. Now, we could use solar energy over a time to split the CO2, but then - wouldn't it be far more efficient to use solar power in the first place and just leave the carbon in the ground where it was in the first place?
Suppose we, instead, sequester the CO2 unprocessed. So - we burn carbon in air to produce CO2 which we then somehow bury. That seems like a good idea. No, wait! Aren't we burying two atoms of oxygen with every atom of carbon? Is that a good idea?
For every molecule of CO2 that we humans produce from the burning of fossil fuels we effectively take at least one molecule of oxygen - O2 - away from atmospheric stocks. As CO2 rises, O2 must fall.
If we bury CO2 then we bury all of that nasty carbon. And twice as much oxygen.
Is that a sensible idea?
Am I missing something?
Do the oxygen fairies put back the oxygen we have consumed that the biosphere can't keep pace with?
From 1991 through 2005, the O2 content of the atmosphere has dropped by 0.00248% (248 per meg) of it's initial amount. The rate is mostly explained by the global combustion of fossil-fuel over this period, although the actual rate is slightly smaller than expected from fossil-fuel alone. The difference evidently reflects a global imballance between photosynthesis and respiration.Quoted text and image source: http://www.esrl.noaa.gov/gmd/.../scripps/...
The O2/N2 data from flasks collected at Mauna Loa are shown in the plot below. Each point is the average of flasks collected on a given day. A smooth trend consisting of a regular seasonal cycle and a long-term trend is fit to the data. The O2/N2 ratio is expressed in per meg units, which express the relative change in the O2/N2 from a standard ratio, multiplied by 1 million.
The graph of O2/N2 should be compared with the Keeling curve below:
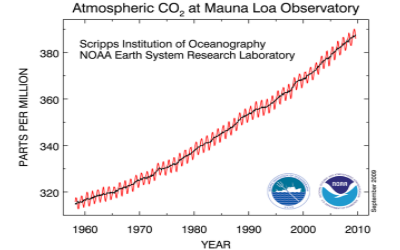
Image source: http://www.esrl.noaa.gov/gmd/
"This is a nightmare, which will pass away with the morning. For the resources of nature and men's devices are just as fertile and productive as they were. The rate of our progress towards solving the material problems of life is not less rapid. We are as capable as before of affording for everyone a high standard of life—high, I mean, compared with, say, twenty years ago—and will soon learn to afford a standard higher still. We were not previously deceived. But to-day we have involved ourselves in a colossal muddle, having blundered in the control of a delicate machine, the working of which we do not understand. The result is that our possibilities of wealth may run to waste for a time—perhaps for a long time."The Great Slump of 1930, John Maynard Keynes.
http://www.gutenberg.ca/ebooks/keynes-slump/keynes-slump-00-h.html
---------------------------
Further reading:
http://blogcritics.org/scitech/article/atmospheric-oxygen-levels-fall-as...
http://sio.ucsd.edu/.../atmospheric+oxygen+levels...






Comments