Techniques scientists currently use to image these transporters of genetic information within cells have several drawbacks, including the need for synthetic RNA or a large number of fluorescent molecules. The fluorescent probes developed at the Georgia Institute of Technology circumvent these issues.
Details of the probe production process and RNA imaging strategy were published in Nature Methods. In addition to Santangelo, Georgia Tech graduate student Aaron Lifland, Emory University associate professor Gary Bassell and Vanderbilt University professor James Crowe Jr. also contributed to this research. This research was funded by new faculty support from Georgia Tech.
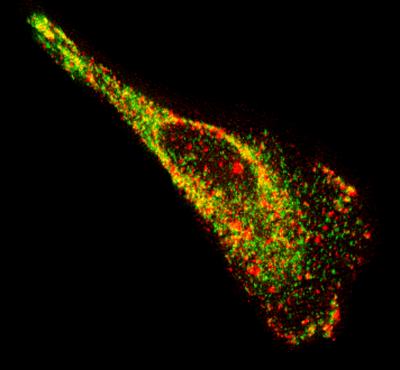
Image of a motile epithelial cell showing native mRNA (red) and a
protein known to bind to mRNA (green). The image reveals a high
concentration of mRNA and proteins along the edges and ends of
the cell. Credit: Georgia Tech Image: Philip Santangelo
"The probes we designed shine bright, are small and easy to assemble, bind rapidly to their targets, and can be imaged for hours. These characteristics make them a great choice for studying the movement and location of RNA inside a single cell and the interaction between RNA and binding proteins," said Philip Santangelo, an assistant professor in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University.
In the study, the probes – produced by attaching a few small fluorescent molecules called fluorophores to a modified nucleic acid sequence and combining the sequences with a protein – exhibited single-molecule sensitivity and allowed the researchers to target and follow native RNA and non-engineered viral RNA in living cells.
"The great thing about these probes is that they recognize RNA sequences and bind to them using the same base pairing most people are familiar with in regards to DNA," explained Santangelo. "By adding only a few probes that would bind to a region of RNA, we gained the ability to distinguish a targeted RNA molecule from a single unbound probe because the former lit up two or three times brighter."
For their experiments, the team used a bacterial toxin to transport the probes into living cells – a delivery technique that when combined with the high affinity of the probes for their targets, required significantly fewer probes than existing techniques. The toxin created several tiny holes in the cell membrane that allowed the probes to enter the cell's cytoplasm.
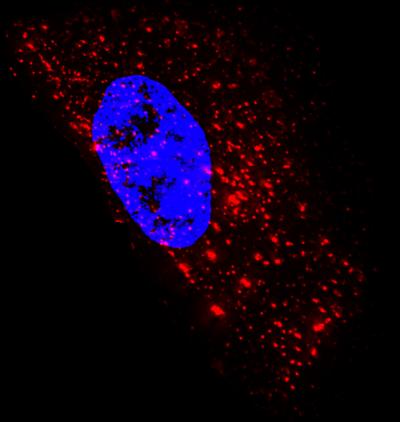
Image of an epithelial cell showing 1,400 native mRNA granules (red) detected using probes designed at Georgia Tech. It is composed of superimposed images taken at several heights above and below the nucleus (blue). Credit: Georgia Tech Image: Philip Santangelo
The researchers tested the sensitivity of conventional fluorescence microscopy to image individual probes inside a cell. Previous studies showed that these techniques were able to image an accumulation of probes inside a cell, but the current study demonstrated that individual probes without cellular targets could be observed homogenously distributed in the cytoplasm with no localization or aggregation.
With single-molecule sensitivity accomplished, the researchers investigated whether they could visualize individual RNA molecules using the probes. To do this, they simultaneously delivered probes designed to target a human messenger RNA (mRNA) sequence region and a probe designed with no target in the human genome. They were able to image unbound probes of both types as well as individual RNA molecules that had attached to the former probes.
The imaging technique also allowed the researchers to observe a process called dynamic RNA-protein co-localization, which is the joining of RNA molecules and RNA binding proteins in a single cell.
"We observed substantial transient interactions between proteins and viral RNA molecules that I don't think had ever been seen before with non-engineered RNA," noted Santangelo. "We saw one of the proteins move into a viral RNA granule and reside within it for over a minute before it was released, and we also saw another protein that appeared to dock with a viral RNA granule."
Santangelo is currently trying to improve the probes by making them smaller and brighter, while also using them to investigate viral pathogenesis and other biological phenomena.
"We are excited to use this imaging strategy to study how single viral RNAs travel from the nucleus of a cell to a virus assembly site, how mRNAs are regulated by location and time, and RNA trafficking in neurons," added Santangelo.






Comments