EBOLA - CLINICAL SYMPTOMS:
Ebola was discovered in 1976 in Zaire and Sudan.
The virus called Ebola takes its name from the Ebola River, which flows into Zaire, where the virus was isolated for the first time.
There are 4 variants of the virus whose differences are due to the type of host they infect: Ebola Zaire, Ebola Sudan, Ebola Tai, that cause disease in humans and primates, and Ebola Reston, which causes the disease only in primates.
During the past 30 years have been recorded periodic epidemics of Ebola in central Africa particularly in Gabon, Uganda, Democratic Republic of Congo, Congo-Brazzaville, Sudan and Angola.
Probably the virus, present in the bats, was manifested later in the great apes and finally in humans. It is transmitted among humans through contact with bodily fluids (blood, vomit, diarrhea ...), which may occur during the care of the sick. This means that, during an outbreak of Ebola, the virus tends to spread to health workers and family members of patients.
The disease presents as flu. Begins with fever, headache, pain in the muscles, then the patient starts to vomit and to have diarrhea.
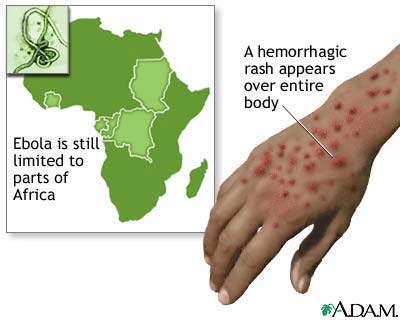
In the final stage in the capillaries of the patient there are tiny clots that produce bruises on the skin and become blisters. After 1-2 days we sees the bleeding arteries and blood flowing from every orifice of the body: eyes, ears, nose. At this point the patient vomits a black liquid, a sign of the disintegration of tissues.
Onset of deadly symptoms occurs between 7 and 14 days incubation, varies from 2 to 21 days, but more commonly between 5 and 10.
The virus has a high transmissibility, and it's transmitted with the direct contact of the fluids of an infected person (blood, secretions, organ transplantation, sexual intercourse), however, although highly contagious, is not transmitted by air.
Up to 90% of people with Ebola die, depending on the strain of the virus.
Crimean fever virus, because of the similarity with the Ebola virus, has been renamed fever virus Crimean-Congo.
There is no specific therapy.
MARPURG - CLINICAL SYMPTOMS:
The virus is named Marpurg, German city, where was isolated for the first time, 1967.
The epidemic occurs for the first time among the staff of a laboratory in the city, with 31 cases and 7 fatalities, while engaged in the cultivation of kidney cells of green monkeys from Uganda, the species Cercopithecus Aethiops, to create vaccines for polio. Subsequently, the disease manifests itself in Frankfurt and Belgrade. The virus appears to be structurally similar to the Ebola, although determines a reaction of different antibodies. The virus Marpurg, reports the magazine "New Scientist" in 2007, has been detected in Gabon in a cave where there were fruit bats. In the same year the virus Marpurg was found in two miners in Uganda.
The incubation period of the disease is about 3-9 days after appearing frontal and temporal headache accompanied by general malaise and myalgias. Characteristic is a high temperature (39-40 ° C), that appears from the first day of illness, followed by a strong and rapid debilitation. About half of patients may complain of conjunctivitis.
In the third day appears watery diarrhea with abdominal pain and cramping, nausea and vomiting, lethargy, and mental changes. In the first week can be manifested cervical lymphadenopathy and the appearance of enanthema of the tonsil and palate. A hallmark is the appearance of a maculopapular non-pruritic rash that appears, usually by the fifth day, on the face and neck and then spreads to the limbs. The hemorrhagic manifestations appeared from the fifth day of illness.
Usually death occurs to cardiovascular collapse due to multiple bleedings. After the first week, the fever begins to subside and then reappear in the twelfth or fourteenth days of illness.
In the second week may also occur: hepatosplenomegaly, facial and scrotal edema.
Usually death occurs mainly between the eighth and ninth to sixteenth days because of continuous bleeding.
HAEMORRHAGIC FEVER VIRUS:
It is unknown whether these viruses have always existed in nature and have always represented a risk to humans or whether these viruses have appeared recently. Were considered as probable zoonotic agents. It is assumed that the filoviruses are endemic mammals in some areas of Africa and who subsequently have adapted to humans. At present knowledge about the origin of these pathogens is still unclear.
There is no antidote or a cure for this disease, so the care that we provide are only supportive and palliative care. Most of our efforts are directed at controlling the epidemic, through the identification of patients and their isolation from the rest of the population. This is being done at this time in the province of Western Kasai.
The incubation period is 3 to 9 days in Marburg virus disease and from 2 to 21 days in the Ebola virus. Symptoms are frequently related to the digestive system (nausea, vomiting, pain, diarrhea) and upper respiratory tract (cough, chest pain, pharyngitis) occurs, then fever associated with myalgia and headache. They may also experience photophobia, conjunctival hemorrhage, jaundice, pancreatitis, and lymphadenopathy. Delirium, sensory disturbances and coma indicate the involvement of the CNS. The next stage is represented by haemorrhage, affecting various organs: nose, intestines, genitals. During the second week of illness, the patient becomes apyretic and start the healing or develops multi organ failure that leading to death.
The diagnosis can be done by the ELISA method (with the identification of both specific antibodies), through the identification of the antigen on immunohistochemical examinations using biopsy resampled to show the virus by electron microscopy and PCR.
The lethality varies between 25 and 90%.
There is currently no specific therapy.
EBOLA NEWS:
Since January 2014 at 30 June 2014, Ebola has affected 759 individuals, with 467 deaths, mostly in Guinea, Sierra Leone and Liberia.
The sperimental drug used for treating ebola is called Zapp, by Mapp Biopharmaceutical Inc. of San Diego. Monoclonal antibodies derived from monkeys infected with the virus, creates an antibody reaction by binding to the surface of the virus. Reports the CDC: "it is too early to know whether ZMapp is effective, since it is still in an experimental stage and has not yet been tested in humans for safety or effectiveness. Some patients infected with Ebola virus do get better spontaneously or with supportive care. It's important to note that the standard treatment for Ebola remains supportive therapy".
The FDA currently does not allow the use of the drug in the USA, in fact Dr. Brantl and Nancy Writebol, infected by the virus, have received therapy in Liberia.
The vaccines currently studied are the TKM-Ebola and BCX4430 but still at a preclinical study. Other interesting vaccine is founded on the vesicular stomatitis virus, VSV, where one of the proteins on its surface is modified to resemble ebola virus, and thus obtain an immune system capable, in contact with the virus, to produce specific antibodies The vaccine was effective in macaques.
There is not currently a suitable therapy therefore the supportive care, such as to the hemorrhagic fever, are the measures of rehydration and administration of paracetamol.
August 2014: WHO, World Health Organization has approved the use of Ebola drugs, although untested, in the West Africa's outbreak.
September 2014: there is not currently Ebola vaccine that the FDA or the CDC considers "safe".
October 2014: FDA has approved the use of experimental drug called brincidofovir producted by Chimerix, a North Carolina biopharmaceutical company.
FDA warns consumers about fraudulent Ebola treatment sold online.
MAY 2018. In the Congo to face the current Ebola epidemic that has reached Mbandaka, a city with one million inhabitants, far more than 80 miles from the site of detection of the original outbreak, over four thousand doses of the experimental VSV-ZEBOV vaccine will be used, revealed effective in limiting the spread of the disease during the previous Ebola outbreak in Guinea, west Africa. Problems are determined by the supply of electricity due to the antidote being stored at a temperature between -60 and -80 degrees.
(rVSV-ZEBOV is a recombinant, replication competent vesicular stomatitis virus-based candidate vaccine expressing a surface glycoprotein of Zaire Ebolavirus We tested the effect of rVSV-ZEBOV , west Africa.
Dr. Ana Maria Henao-Restrepo MD et al Lancet https://doi.org/10.1016/S0140-6736(16)32621-6).






Comments