NASA scientists reproduced uracil in a lab under conditions found in space, according to Astrobiology. Uracil is one of the components of the genetic code that makes up ribonucleic acid (RNA); RNA is mainly known for its role in protein synthesis. In other words, NASA was able to create a building block of life in the lab.
 Jesus bringing the divine hate.
Jesus bringing the divine hate.But why would that piss off Jesus? First, a brief lesson in nucleic acids.
Purines and Pyrimidines
The three main needs of processes within the cell are nucleic acids, proteins, and membranes. Nucleic acids are, very simply, molecules that carry genetic information in all living cells and viruses. The two heavyweights are deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). At its most basic, DNA is the blueprint, the card catalog, the information storage unit, whereas RNA is the delivery truck, getting the information to the folks in the cell that make proteins.
DNA is generally composed to two chains that wrap around each other (a double helix), and RNA is typically a single chain. The chain "links," or bases, in DNA and RNA are almost identical. Both have bases called cytosine, guanine, and adenine. However, the fourth base for DNA is thymine, while the fourth base for RNA is uracil.
Guanine and adenine are members of the purine group, which share a common foundation of two rings (see below). Caffeine is also a purine.

Cytosine, thymine and uracil are all pyrimidines, which all share the common foundation of a single ring (see below).
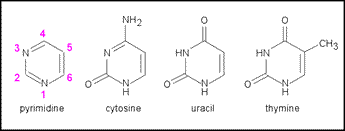
If you look closely, you can see uracil and thymine are almost the same molecule, except thymine has an attached methyl group (CH3).
Why do we care about purines and pyrimidines? Well, they're the lego pieces in the building blocks of life. The key question is, where do they come from? Here's the kicker - some of it comes from space.
"The fault, dear Brutus, is not in our stars, / But in ourselves, that we are underlings."
Meteorites can contain a soup of organic compounds, including pyrimidines and purines. The problem is that early Earth life was a lot like Dick Cheney - hostile, volatile and full of hot air. Not exactly conducive to survival of stable nucleotides (and formation of amino acids and proteins).3 Plus, space itself isn't a big warm and fuzzy nurturing environment. So, how did pyrimidines survive out there?
"Molecules like pyrimidine have nitrogen atoms in their ring structures, which makes them somewhat whimpy. As a less stable molecule, it is more susceptible to destruction by radiation, compared to its counterparts that don’t have nitrogen," aid Scott Sandford, a space science researcher at Ames. "We wanted to test whether pyrimidine can surviveResearchers thought that if pyrimidine made it to Earth in meteorites it had to survive somehow - maybe interstellar dust shielded the compounds from radiation, which would blast it to bits. And once it was in the dust it would freeze anyway, further protecting it.
in space, and whether it can undergo reactions that turn it into more complicated organic species, such as the nucleobase uracil."
During their experiment, they exposed the ice sample containing pyrimidine to ultraviolet radiation under space-like conditions, including a very high vacuum, extremely low temperatures (approximately - 340 degrees Fahrenheit), and harsh radiation.The researchers thought the experiment could be "an important linking step between what goes on in space and what fell to Earth early in its development."
They found that when pyrimidine is frozen in water ice, it is much less vulnerable to destruction by radiation. Instead of being destroyed, many of the molecules took on new forms, such as the RNA component uracil, which is found in the genetic make-up of all living organisms on Earth.
So, the Earth formed from star dust, and later meteorites and comets delivered even more materials to our planet that could have formed primitive life's building blocks. Now NASA has figured out how to replicate this in a lab.
Nowhere in that sentence do I read "God created the heavens and the earth," but maybe I'm missing something. I checked Genesis, but couldn't find anything. Playing God, are we? Horning in on Jesus' game? Well, if Jesus comes knock, knock, knocking at NASA's door, hopefully it's Buddy Christ and not cyborg pirate ninja Jesus.
1 Or, rather, creationists in Jesus' name.
2 Clearly NASA committed a mortal sin, versus the lesser venial version. Mortal sins are knowing and willful violation of God's law. Venial sins, on the other hand, merely injure our friendship with God (although you can still pal around and maybe get some pizza later).
3 What could possibly survive during those early millennia, however, were molecules like polycyclic aromatic hydrocarbons (PAH). In a paper in Astrobiology, authors suggested that the "aromatic material can be used as a container, as a metabolic unit, and as a genetic information carrier. We think that aromatic material can be used for all three requirements for life."






Comments