Drs. Fred Cross and Eric Siggia have produced a steady stream of outstanding systems-level studies of one of the most important biological oscillators: the cell division cycle. I'll have more later today on their fascinating new paper on phase-locking the cell cycle, but in the mean time, check out their recent
methods paper, which has some great movies of live-imaged yeast cells doing various cell cycle tricks, visualized with fluorescent proteins:
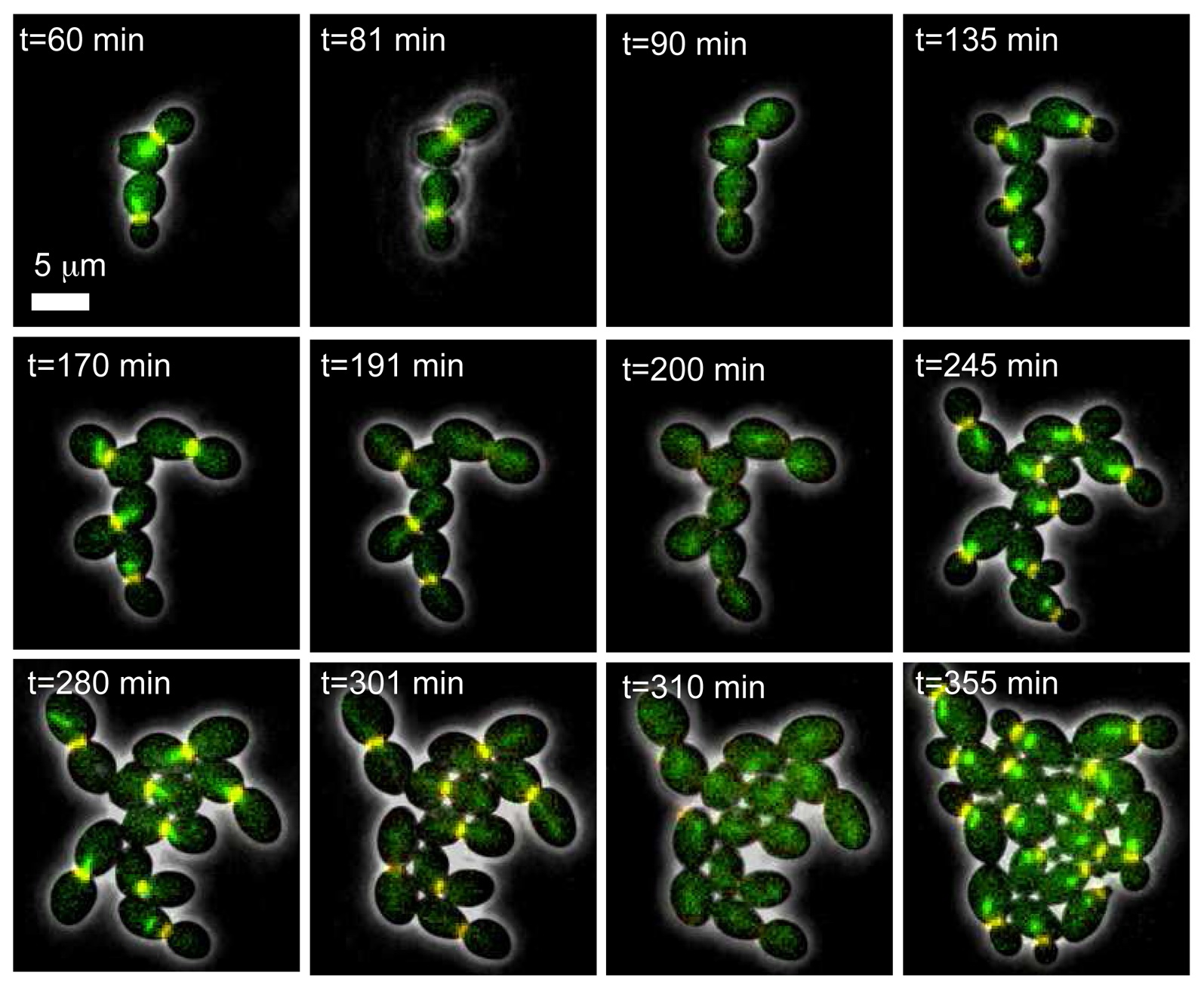
Here, we report a novel microfluidic setup for yeast that uses a dialysis membrane to separate the cells from the external flow. It provides much better confinement and planar growth than with gel pads and therefore allows high resolution imaging of single cells for more than 8 division cycles. Dense fields of cells can be segmented from a phase image, leaving the fluorescent channels free. Media can be rapidly changed with no visible movement in the growing colony. We have studied the induction of the regulatable GAL1 and MET3 promoters with their respective metabolites, and have shown that externally controlled pulses of gene expression can be achieved with a 10 minutes resolution using the MET3 promoter. These experiments combined with modeling have allowed a precise quantitative characterization of single-cell responses of these promoters to induction... The coupling of imaging with a tight control over gene expression will provide accurate and tightly controlled data for any dynamic process with a fluorescent or morphological readout.






Comments