In Artificial Hairy Surfaces with a Nearly Perfect Hydrophobic Response, the authors took their inspiration from spiders, particularly the diving spiders that can gather a bubble of air which they carry with them underwater. Whereas others had previously tried uniform nanofibre coatings, these authors noted that spider hairs are very heterogeneous, both large and small, and straight and curved.
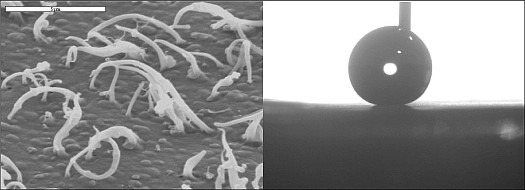
They generated the artificial hairy surface on a polypropylene surface (picture left) by moulding against a polycarbonate membrane. Whereas on plain polypropylene, water droplets will stand like domes, on the hairy surface the water droplets stand up in a full spherical shape (picture right). This indicates a completely hydrophobic surface, giving minimum drag, which we may soon find on speedboats and Olympic swimwear.
This was drawn to my attention through this article in Live Science: New Material Patterned After Spider Hair Refuses to Get Wet.
* * * * * * *
However, my most reliable source of inspiration is the magazine Chemistry World, which drops through my door every month. In the latest number is a news item based on this next paper: Directional water collection on wetted spider silk.
So, time for a double-take. Spider skin keeps one dry, spider silk collects water! It reminds me of Aesop’s fable about the centaur, alarmed at the man who blew on his hands to warm them, and on his porridge to cool it. How could he stay in the house with a creature that could blow hot and cold from the same mouth?
Well, we know that water can stick to spider silk, seeing the dewdrops glistening on autumn webs. The picture below is part of a lovely Wikimedia picture.
.jpg)
The authors show that in one spider at least this water collecting ability is due to
structural features [which] result in a surface energy gradient between the spindle-knots and the joints and also in a difference in Laplace pressure, with both factors acting together to achieve continuous condensation and directional collection of water drops around spindle-knots.
They have used this to generate artificial webs for moisture collection, with a view to capturing water in places which may be experiencing reduced rainfall as a result of climate change.
Note: Laplace Pressure is a thermodynamic phenomenon which causes large droplets or bubbles to cannibalize small ones. (This tutorial explains the importance of this phenomenon in regard to premature babies.)
* * * * * * *
Now one may get fed up with all those companies that keep sending you emails advertising their products. However, the regular missal sent out by NIL Technology, their NILT Nano Newsletter, is full of all sorts of goodies they’ve scoured from the news. Here’s another non-wettable, this time without the spider.
In Morphology of Air Nanobubbles Trapped at Hydrophobic Nanopatterned Surfaces, the surface is silicon, which they coat with a block copolymer which forms a hexagonal nanopattern. They then use this coating as a mask to etch the honeycomb-like pits as shown in the picture below at right. According to thermodynamics as generally understood, the air trapped in the tiny pits ‘didn’t ought to be there’, since the tiny bubbles ought to disappear through Laplace pressure (again.) The authors discuss this in terms of a dynamic equilibrium, but I think the jury is still out on this one.

They envisage that
The nanotextured surfaces described here . . . may find applications, for example, in micro- and nanofluidics as a means for reducing the friction of liquid flow into small channels.
What is common to all three papers here is that the authors are not acting as “just” physicists or “just” chemists or even arachnologists. Although one can’t know everything, an overview of what is going on in science seems to be the order of the day.





Comments