The invasive nature of cancers in general, and malignant gliomas in particular, is a major clinical problem rendering tumors incurable by conventional therapies.
Using a novel invasive glioma mouse model established by serial in vivo selection, new research has identified the p75 neurotrophin receptor (p75NTR) as a critical regulator of glioma invasion. Through a series of functional, biochemical, and clinical studies, it was found that p75NTR dramatically enhanced migration and invasion of genetically distinct glioma and frequently exhibited robust expression in highly invasive glioblastoma patient specimens.
It was also found that p75NTR-mediated invasion was neurotrophin dependent, resulting in the activation of downstream pathways and producing striking cytoskeletal changes of the invading cells. These results provide the first evidence for p75NTR as a major contributor to the highly invasive nature of malignant gliomas and identify a novel therapeutic target.
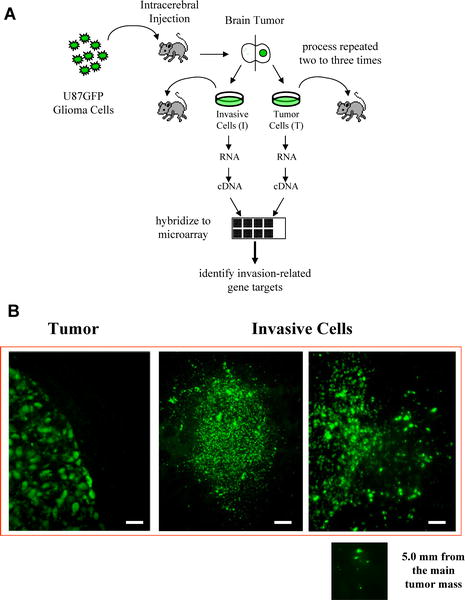
(B) Brains of SCID mice implanted with either tumor (left) or invasive (center and right) glioma cells. GFP visualization reveal the well-circumscribed border of the reimplanted tumor cells, with no tumor cells being detected away from the main tumor mass (left). This is in sharp contrast to the highly invasive border of the invasive tumors, where isolated small groups of glioma cells are found throughout the brain (center and right). Scale bars on GFP images represent 125 μm (center) and 62 μm (right and left). From: The p75 Neurotrophin Receptor Is a Central Regulator of Glioma Invasion Johnston ALM, Lun X, Rahn JJ, Liacini A, Wang L, et al. PLoS Biology Vol. 5, No. 8, e212 doi:10.1371/journal.pbio.0050212
Gliomas are highly malignant and invasive tumors with tendrils that extend far from the primary tumor site, rendering conventional therapies ineffective and leading to an invariably poor prognosis.
In the new study published online this week in PLoS Biology, Angela Johnston, Donna Senger, and colleagues at the University of Calgary injected immunocompromised mice with human gliomas and compared invasive cells (which left the primary tumor site) to noninvasive cells (which remained at the site of injection) in order to understand the molecular mechanisms underlying this invasive behavior.
They identified the neurotrophin receptor p75NTR, which normally functions during development to induce neurite outgrowth and promote neuronal cell death, as an important regulator of glioma invasion. The researchers present the first evidence that this neurotrophin receptor can also be a potent mediator of glioma invasion, and they show that the expression of this receptor is sufficient to impart a dramatic invasive behavior on genetically distinct tumors.
These data highlight a previously unknown function of this receptor and suggest it may be a novel therapeutic target in the treatment of this devastating cancer.
Johnston ALM, Lun X, Rahn JJ, Liacini A, Wang L, et al. (2007) The p75 neurotrophin receptor is a central regulator of glioma invasion PLoS Biol 5(8): e212. doi:10.1371/journal.pbio.0050212.






Comments