Collagen, the most abundant protein in the body, is widely used to build scaffolds for tissue engineering because it is biocompatible and biodegradable. Collagen is, however, hard to work with in its natural form because it is largely insoluble in water, and common processing techniques reduce its strength and disrupt its fibrous structure.
Tufts University School of Engineering researchers have developed a new method for fabricating collagen structures that maintains the collagen's natural strength and fiber structure, making it useful for a number of biomedical applications.
The new technique, called bioskiving, creates collagen structures from thin sheets of decellularized tendon stacked with alternating fiber directions that maintain much of collagen's natural strength.
Bioskiving does not dilute collagen's natural properties, says Qiaobing Xu, assistant professor of biomedical engineering, and inventor of the new technique. "Our method leverages collagen's native attributes to take advantage of the well-organized micro/nanostructures that nature already provides," he says.
Slice, Stack, and Roll
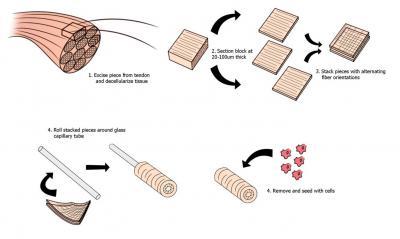
This inforgraphic depicts the new bioskiving technique developed at Tufts. Credit: Qiaobing Xu
In their research, Xu and Kyle Alberti, a Ph.D. student in Xu's lab, cut small sections of collagen from bovine tendons. Using a specialized detergent, the researchers decellularized the sections, leaving intact only the extracellular collagen matrix made of bundles of aligned collagen nanofibers.
Xu and Alberti sliced the sections into ultra-thin sheets using a microtome, and then stacked 10 slices, crisscrossing the sheets so that the fibers in one ran perpendicular to those above and below it. This process produced a scaffold material with tensile strength stronger than constructs made using common processing techniques, Xu notes.
The researchers also created tubular scaffolding by rolling layers of collagen sheets around Teflon-coated glass rods. The sheets were layered so that fibers ran along the length and the circumference of the rods. This process yielded tubes that were found to be stronger than similar tubes made of reconstituted collagen. They also maintained their highly aligned fiber structure.
"Alignment gives the scaffold the ability to guide the direction and orientation of cell growth," says Xu, who also has a faculty appointment at Tufts School of Medicine, "This capability is beneficial for tissue engineering applications where biocompatibility and the ability to guide unidirectional nerve growth are both desired, such as prosthetic or tissue engineering-based blood vessels or nerve conduits."
Published in Advanced Healthcare Materials.





Comments