A team of researchers headed by Prof Dr. Nicole Frankenberg-Dinkel at Ruhr-University Bochum have revealed similarities and differences in the assembly of the light-harvesting machinery of the cryptophyte Guillardia theta compared to cyanobacteria and red algae.
Cryptophytes: Matryoshka dolls of the waters
Unlike traditional eukaryotic cells – i.e. all cells with a nucleus – cryptophyte cells resemble a Russian doll in the form of an alga within an alga. They originated from a eukaryotic cell that engulfed and integrated a red alga. Thus, cryptophytes have acquired the ability to perform photosynthesis. Just like their red algal ancestors, cryptophytes utilise not only the green pigment chlorophyll for harvesting light, but have also the capability to use green light being missed by chlorophyll.
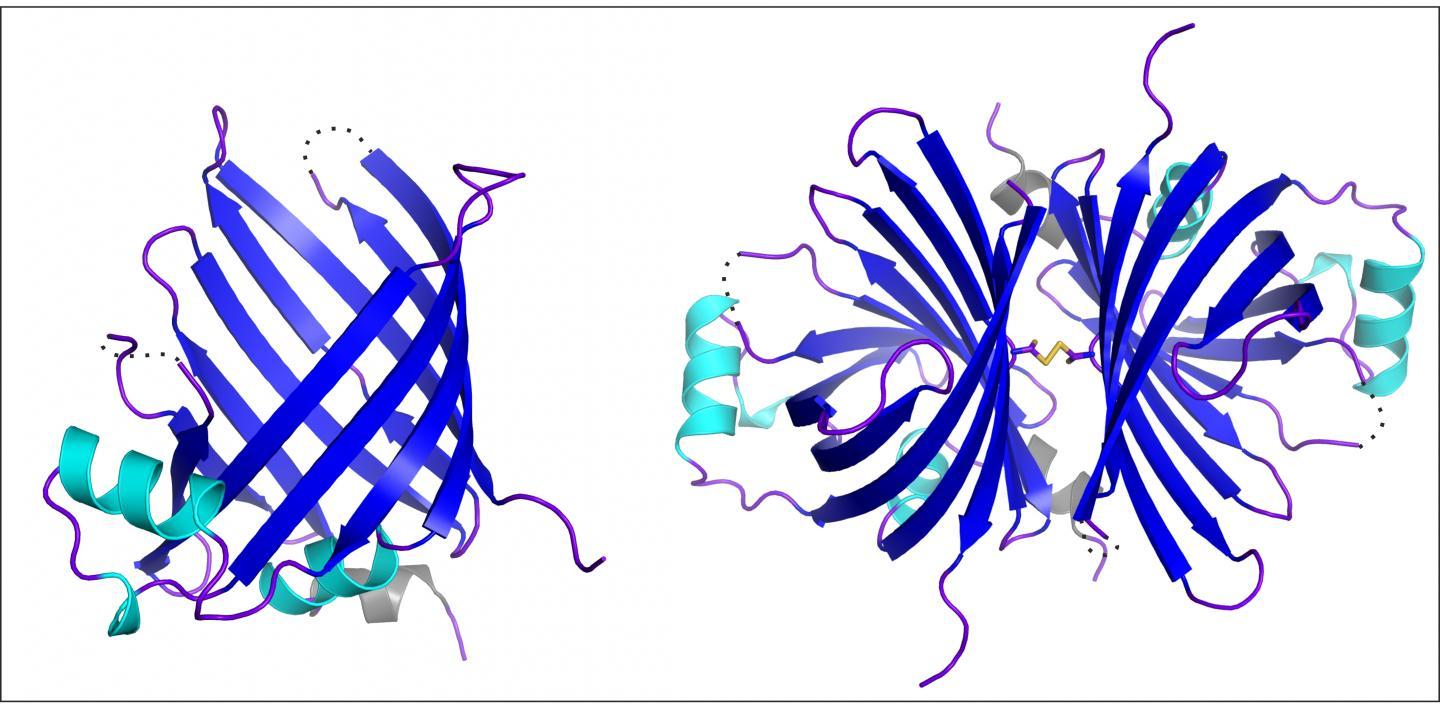
This image shows the atomic structure of the GtCPES protein in its monomeric (left) and dimeric (right) form, which has been solved by X-ray structural analysis. GtCPES forms a barrel-like structure, which is closed at the bottom and open at the top. Inside the barrel, molecules such as phycoerythrobilin can be bound and transported. Credit: Kristina Overkamp
This is due to blue or red-tinted proteins – so-called phycobiliproteins. After integrating the red alga, its genome was reduced over time and combined with the host cell's genome. During this process, a number of modifications on genetic, biochemical and physiological levels took place, which facilitated adaptation to new ecological niches.
Although the algae maintained the basic principle of photosynthesis, their light-harvesting phycobiliproteins were largely modified and greatly differ from their cyanobacteria and red algae ancestors.

A schematic of the complex construction of a cryptophyte cell. The barrel-like docking enzyme GtCPES is found in an inner compartment, the chloroplast stroma. Here, the enzyme is responsible for the transport and docking of the magenta-colored pigment phycoerythrobilin to the phycobiliprotein structure. Credit: Kristina Overkamp
Protected pigment transport in a barrel
In many respects, the way light-harvesting works in Guillardia theta has not yet been understood. RUB researchers have now for the first time gained insight into the complex biosynthesis of cryptophyte phycobiliproteins. "Guillardia theta has obviously combined tried-and-tested and novel synthesis methods and enzymes," says Prof Dr Nicole Frankenberg-Dinkel. The biosynthesis of the magenta-coloured light harvesting pigment phycoerythrobilin is similar to that of cyanobacteria.
Docking of pigments on the phycobiliprotein structure, on the other hand, is performed by both known and novel enzymes. Prof Frankenberg-Dinkel's team of researchers succeeded in characterising one of these docking enzymes, GtCPES, in biochemical and structural terms. In collaboration with Dr Raphael Gasper-Schönenbrücher from the work group Protein Crystallography, headed by Prof Dr Eckhard Hofmann, they solved the atomic structure of GtCPES. GtCPES is shaped like a barrel that is sealed only at the bottom end and is open at the top to only let in the magenta-coloured pigment phycoerythrobilin.
The barrel's function is to shield the sensitive pigment from external influences while it is being transported to its destination, i.e. to the apo-phycobiliprotein. Thanks to the barrel top's structural properties, the transfer of the pigment is carried out in the correct orientation to a clearly defined spot of the target protein surface.
Published in The Journal of Biological Chemistry. Source: Ruhr-University Bochum






Comments