Dyes absorb photons from sunlight and generate a charge in the form of electrons, which are captured first by a semiconducting titanium oxide layer deposited on a current collector before flowing back to the counter electrode through another current collector. Progress has been made in the manufacture of DSCs that incorporate an iodine-based electrolyte, but iodine tends to corrode metallic current collectors, which poses a challenge for its long-term reliability. Iodine electrolyte also has the unfortunate tendency to absorb light in the visible wavelengths, which means fewer photons could be utilized.
Single-wall nanotube arrays, grown in a process invented at Rice, are both much more electroactive and potentially cheaper than platinum, a common catalyst in DSCs, said Jun Lou, a materials scientist at Rice. In combination with newly developed sulfide electrolytes synthesized at Tsinghua, they could lead to more efficient and robust solar cells at a fraction of the current cost for traditional silicon-based solar cells.
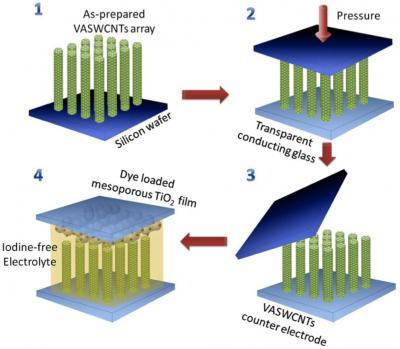
Arrays of vertically aligned single-walled carbon nanotubes (VASWCNTs) grown at Rice University are key to making better and cheaper dye-sensitized solar cells, an alternative to more expensive silicon solar cells. The arrays are transferred to conducting glass, topped with a second electrode of titanium oxide and surrounded by iodine-free electrolyte developed at Tsinghua University. Credit: Lou Lab/Rice University
Tsinghua researchers decided to try a noncorrosive, sulfide-based electrolyte that absorbs little visible light and works well with the single-walled carbon nanotube carpets created in the Rice lab of Robert Hauge, a co-author of the paper and a distinguished faculty fellow in chemistry at Rice's Richard E. Smalley Institute for Nanoscale Science and Technology.
"These are very versatile materials," Lou said. "Single-walled carbon nanotubes have been around at Rice for a very long time, and people have found many different ways to use them. This is another way that turns out to be very well-matched to a sulfide-based electrolyte in DSC technology."
Both Rice and Tsinghua built working solar cells, with similar results. They were able to achieve a power conversion efficiency of 5.25 percent – lower than the DSC record of 11 percent with iodine electrolytes and a platinum electrode, but significantly higher than a control test that combined the new electrolyte with a traditional platinum counter electrode. Resistance between the new electrolyte and counter electrode is "the lowest we've ever seen," Lou said.
There's much work to be done, however. "The carbon nanotube-to-current collector still has a pretty large contact resistance, and the effects of structural defects in carbon nanotubes on their corresponding catalytic performance are not fully understood, but we believe once we optimize everything, we're going to get decent efficiency and make the whole thing very affordable," Lou said. "The real attraction is that it will be a very low-cost alternative to silicon-based solar cells."
Citation: Feng Hao, Pei Dong, Jing Zhang, Yongchang Zhang, Phillip E. Loya, Robert H. Hauge, Jianbao Li, Jun Lou&Hong Lin, 'High Electrocatalytic Activity of Vertically Aligned Single-Walled Carbon Nanotubes towards Sulfide Redox Shuttles', Scientific Reports 2, Article number: 368 doi:10.1038/srep00368






Comments