Ever since the 1985 discovery of C60, this molecule, with its perfect geodesic dome shape has fascinated scientists, physicists, and chemists alike. Like a soccer ball, the molecule consists of 20 carbon hexagons and 12 carbon pentagons. The electronic properties of C60 are very unusual, and there is a massive research effort toward integrating it into molecular scale electronic devices like transistors and logic gates.
To do this, researchers need to know how the molecule forms bonds with a metal substrate, such as silver, which is commonly used as an electrode in devices. Now, Hsin-I Li, Renee Diehl and colleagues have determined the geometry of C60 on a silver surface using a technique called low-energy electron diffraction.
They find that the silver atoms rearrange in such a way – namely, by forming a 'hole' beneath each C60 molecule - that reinforces the bonding between the carbon structure and the silver surface.
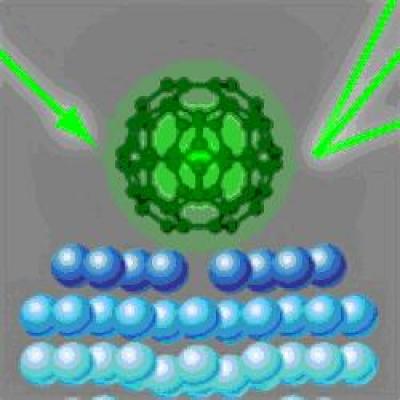
The complete structure of C60 molecules on a silver surface with electron diffraction. Credit: Image copyright American Physical Society [Illustration: Alan Stonebraker after H. I. Li et al.]
The measurements push the limits of surface science because the molecules and the re-arrangement of the underlying silver atoms are quite complex. The measurements thus open the door to studies of a large number of technologically and biologically important molecules on surfaces.






Comments