Hepatocellular carcinoma (HCC) is the second leading cause of cancer-associated death worldwide, due to the difficulty in treating this cancer using conventional chemotherapeutic drugs such as doxorubicin, epirubicin, cisplatin, 5-fluorouracil or etoposide.
A group in Asia believes this may be because medicines are not able to reach liver tumor cells in sufficient levels without harming to the rest of the body. They believe alternative drug treatment options that are able to target the tumor tissues, without inducing toxicity in other parts of the body, may be the solution, so Professor Taeghwan Hyeon at Seoul National University and Professor Kam Man Hui at the National Cancer Center Singapore, screened a library containing hundreds of natural products against a panel of HCC cells to search a better drug candidate.
Their screen found that a compound named triptolide, a traditional Chinese medicine isolated from the thunder god vine (Tripterygium wilfordii (Latin) or lei gong teng (Chinese), was more potent than current therapies and cite other claims that triptolide is effective against other malignant cancers including pancreatic, neuroblastoma and cholangiocarcinoma. However this excitement was tempered when the drug was administered to mice as the increased potency was coupled with increased toxicity as well.
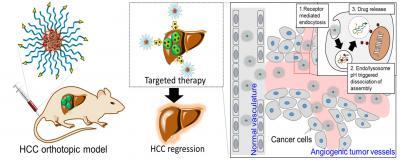
Nf-Trip for Hepatocellular Carcinoma (HCC) targeted therapy. Credit: © Institute for Basic Science
Maximizing potency, mitigating toxicity
Hyeon et al. alleviate the toxic burden by increasing the specific delivery of the drug to the tumor using a nanoformulation. The designed formulation was a pH-sensitive nanogel coated with the nucleotide precursor, folate. The researchers began by esterfying the polymer pluronic F127 with folate to make the coating material. They then polymerized β-benzyl-L-aspartate N-carboxy anhydride to make the core material pH-sensitive due to repulsive forces upon protonation under acidic conditions.
"The combination of the two polymers forms a core/shell structured nanoparticle in water," explains Hyeon. "We loaded triptolide into the hydrophobic core to produce a kind of drug-nanogel."
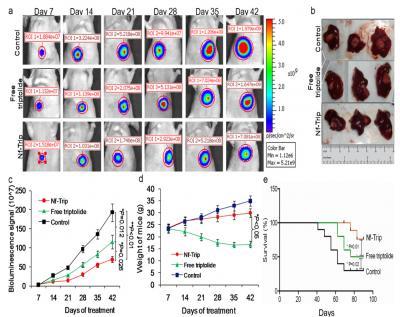
This image proves that Nf-Trip suppresses tumor growth of HCC orthotopic model. Credit: © Institute for Basic Science
A tumor model of folate-overexpressing HCC was then used to examine the effect of the nanogel formulation versus the free drug. As expected, the nanogel triptolide showed increased tumor accumulation and uptake into the tumor cells where the decreasing pH efficiently triggered release of the entrapped triptolide. The result was as hypothesized: In experiments on mice with HCC, the team found that its coated triptolide accumulated in the inflamed tumour tissues.
Once there, the folate-targeted ligand enhances the HCC cells to take up the anticancer drug. Since the fluid inside tumour cells is more acidic (with a pH of around 6.8) compared to normal tissue (which has a pH of about 7.4), the drop in pH causes the coating to fall apart, and release the pure form of the triptolide, which then destroys the tumor cells, showing greater efficacy against the tumor and decrease the overall toxicity.
The mechanism of action of Nf-Trip-FR+ represents an auspicious therapeutic approach
While these initial proof-of-concept studies have been promising, many drugs fail to become an IND (Investigational New Drug); fewer still effectively replicate their results in human trials. However, a felicitous discovery occurred while the researchers were examining the mechanism of triptolide's activity. Researchers at the National Cancer Center Singapore ran a profile on the effects triptolide had on protein expression in a variety of HCC cells. From this they learned triptolide primarily reduced the levels of two proteins, AURKA and CKS2, although the mechanism is still not known. The researchers then cross-checked these proteins against a clinical database of HCC patients and found an increased expression of these proteins correlates with the aggressiveness of the cancer. Thus it is hoped the negative effect triptolide has on these proteins could prove beneficial in terms of clinical outcomes when this drug finally becomes accepted for clinical studies in cancer patients.
The present work is detailed in ACS Nano.






Comments