Synthetic biology can provide a faster and cheaper route, says Jay Keasling, a chemical engineer with joint appointments at Berkeley Lab and UC Berkeley, who led the development of a microbial-based method of producing artemisinin. Fermenting artemisinin via engineered microbes, such as yeast, can be done at far lower costs than , making microbial-based artemisinin a much cheaper but equally effective treatment. Restricting access to this technology to responsible manufacturers who will bundle artemisinin as part of an anti-malarial drug "cocktail" rather than selling it as a monotherapy should delay or even prevent malaria parasites from developing resistance - recently, there have been reports of malaria parasites in West Africa showing some signs of resistance to artemisinin.
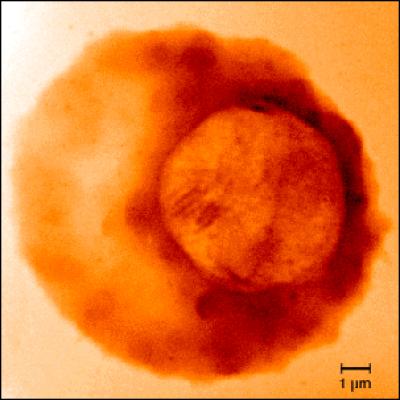
As shown in this X-ray image taken at Berkeley Lab's Advanced Light Source, the malaria parasite develops inside red blood cells, where it accumulates iron. It is vulnerable to the oxygen-based free radicals released by a powerful but scarce antimalarial drug known as artemisinin. Credit: Lawrence Berkeley National Laboratory
"The problem has been that some manufacturers have sold artemisinin as a monotherapy rather than as a co-therapy as is recommended by the World Health Organization," said Keasling. "Any drug that is used as a monotherapy raises the possibility of microbes developing resistance to it. Right now artemisinin is grown by farmers all over the world and sold to anybody. Through the synthetic biology technique, access to the cheapest artemisinin can be restricted to manufacturers who agree to sell it as part of a co-therapy drug."
Keasling is also CEO of the Joint BioEnergy Institute (JBEI), one of three U.S. Department of Energy Bioenergy Research Centers.
"Synthetic biology is somewhat like building a computer from the off—the-shelf parts," Keasling said in his talk. "You have a knowledge base, off-the-shelf components, a system for assembling these parts and an idea as to what you want to do."
For the microbial synthesis of artemisinin, the idea started with malaria, a disease first described in 4 B.C. by Hippocrates that continues to claim the lives of more than a million victims each year, most of them children. The complex life-cycle of Plasmodium falciparum, the parasite that carries malaria, makes it impossible to eradicate the disease. Treatment is the only option and the most effective current treatment is artemisinin, which releases high doses of oxygen-based free radicals that destroy the Plasmodium parasite while it is inside a red blood cell. The cost of extracting artemisinin from wormwood trees, which only produce the drug under a narrow set of agricultural and climatological conditions, or manufacturing it entirely through chemical synthesis, is so high that impoverished populations in Africa and South America who need it the most cannot afford it.
In 2002, Keasling and members of his research group, using the tools of synthetic biology, set out to engineer a microbe that would perform most of the chemistry needed to make artemisinin in order to substantially reduce production costs. In 2003, they reported their first success. By transplanting genes from yeast and from the sweet wormwood tree into E. coli bacteria and then bypassing the E. coli's metabolic pathway and engineering a new one based on the mevalonate pathway in yeast, they were able to induce the bacteria to produce amorphadiene, a chemical precursor to artemisinin.
"Initially production was low, but we have used gene re-synthesis and other techniques to improve the yield of amorphadiene in E. coli by a million fold from where started," Keasling said.
In 2004, Keasling received a $42.6 million grant from the Bill and Melinda Gates Foundation, through the Institute for OneWorld Health, a San Francisco-based nonprofit pharmaceutical company, to further develop his microbial artemisinin technology. A bio-tech start-up company, Amyris Biotechnologies, Inc., which is based in Emeryville, licensed the technology. In 2006, the collaboration reported the use of synthetic biology techniques to genetically engineer a strain of yeast (Saccharomyces cerevisiae) that was capable of producing high levels of artemisinic acid, the immediate precursor to artemisinin.
"Given the existence of known, relatively high-yielding chemistry for converting artemisinic acid to artemisinin or any other derivative that might be desired, microbial produced artemisinic acid is a viable source of this potent family of anti-malarial drugs," said Keasling when the yeast breakthrough was first announced.
The engineering of the yeast was a three-step process in which Keasling and his group first created a new metabolic pathway in the yeast, similar to the one created in E. coli, then introduced bacterial and wormwood genes into the yeast's DNA that interacted with the yeast's own genes to produce amorphadiene. Finally, they cloned the gene from the wormwood tree that produces the enzyme P450, which the plant uses to convert amorphadiene to artemisinic acid, and expressed it in the amorphadiene-producing yeast strain.
"We got it right the first time we tried it," Keasling said at his AAAS talk. "This is not unprecedented in the literature but it is really memorable when it happens to you!."
Last year, Keasling and his collaborators formed a new partnership with Sanofi-aventis, a leading pharmaceutical company based in France, with the goal of mass-producing low-cost microbial-based artemisinin by 2010.
"Over the long run, we expect to be able to reduce the production costs by an order of magnitude, which will fulfill our goal of meeting and beating the current cost of plant-derived artemisinin," Keasling said. "The concept is to produce artemisinin cheaply and sell it to companies that will produce the right kinds of co-therapies and enable them to compete with companies that are trying to market artemisinin as a monotherapy."
Keasling said that the same synthetic biology techniques used to make the microbial-based artemisinin should be equally effective in the drive to produce a next generation of biofuels – carbon-neutral transportation fuels derived from plant biomass.
"Artemisinin is hydrocarbon and we built a microbial platform to produce it. We can remove a few of the genes to take out artemisinin and put in a different hydrocarbon to make biofuels."
As Keasling is fond of saying, "With the tools of synthetic biology, we don't have to just accept what Nature has given us."






Comments