We learned from the prior blog that the Sun is much too bright for normal viewing with our sensitive eyes. A white “color” results when we observe objects that are both extremely bright and bright at all or most visible wavelengths. Please allow me to elaborate a little more on this issue.
[Hang in there.]
This white result can be quite deceptive since its true color could be any color once we attenuate the object’s intensity to a light flux conducive to our normal eyesight. The reason for this blinding effect is due to the behavior of our color receptors (color cones) when they are subjected to excessive light. We have about 6 or 7 million of these cones in each retina. Our cones come in three types – surprisingly, there are four types of cones for some birds – and they are often designated as our red, green, and blue color cones due to their spectral sensitivity to the respective wavelengths of light. Our trichromatic eyesight gets quite complicated since there is much overlap of their sensitivity with one another. Fortunately, the brain handles it amazingly well.
[BTW, many animals are dichromatic. Whitetailed deer, for instance, have just two color cones: essentially, blue and yellow. It seems likely they are capable of seeing into the UV band. Regardless, these dears can probably see a somewhat bright sky shining upon hunters as hunters stumble through their darkness.]
If all these color cones become saturated due to excessive amounts of light shining upon them, then the brain will produce a white sensation for the bright object being observed. This excessive flux eliminates any chance for the brain to determine the true color of the object; the objects true color is hidden behind the blaze.
This blinding white result is what happens when we observe the Sun with the naked eye when the Sun is directly overhead, or, worse, when we look at it from space without the safety visor pulled-down from atop our space helmet. Once the Sun nears the horizon, where direct sunlight must pass through much more atmosphere to reach us, the Sun's intensity becomes more comfortable to observe. But when it is high in the sky, it is dangerously bright to observe.
This same problem of excessive light flux also applies to electronic light sensors. The Hubble Space Telescope, for instance, is programmed to avoid looking in any region within about 50 degrees of the Sun, else damage can occur to its very sensitive instruments. The HST also avoids pointing near either the Earth or the Moon for the same reason, though it can point closer to these dimmer objects. [Actually, the HST is capable of imaging the Moon, but care must be taken.]
With the exception of the cooler stars, black holes, and neutron stars, all stars emit light at all wavelengths of the visible spectrum. Their output is known as a Planck or blackbody distribution. They are not perfect blackbody radiators, but they are close. For this reason, our color cones will be activated in proportion to the relative spectral energy distribution (SED) of the light emitted from the star. The hotter stars will excite our blue color cones proportionately more so and will cause us to see these stars as blue-white. Red stars are much stronger in red than any other wavelength, thus we will see these as red or orange stars. This assumes, once again, that we are not blinded due to excess intensity from these colorful stars.
Whew, that was a lot of dry stuff, and heliochromology is supposed to be fun. Ok, gander at this:
That is the Sun's spectrum upon my son's face. [Thanks Matt!!] All the colors are vividly present. Notice, however, how very little yellow light is apparent in this spectrum created by using a narrow slit and an F2 prism.
So, what about the Sun’s true color? Well, let’s now look at the hard data: spectral irradiance:
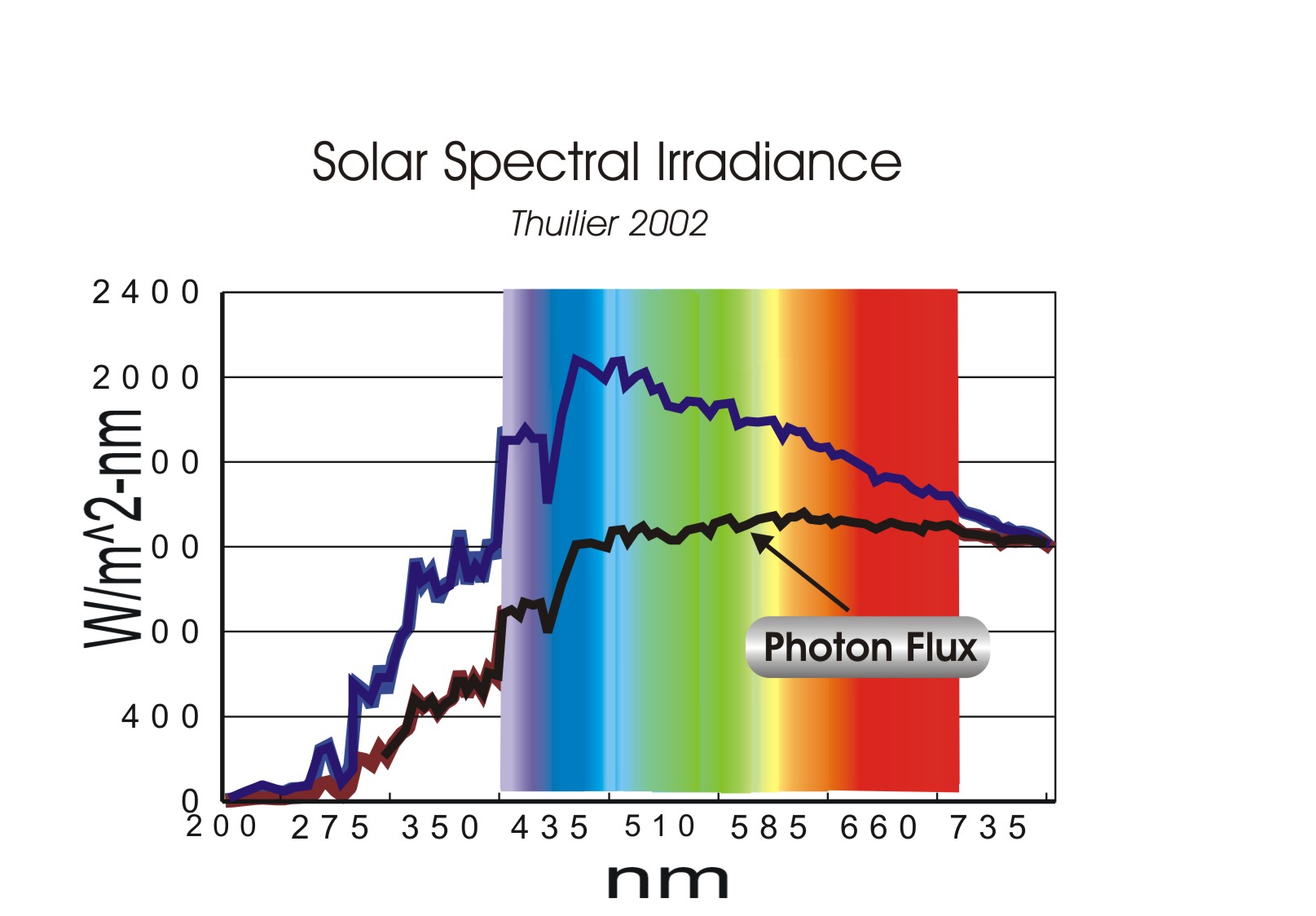
This data comes from just one of hundreds of data sets taken of the Sun from space, AM0. The background color is added to show a reasonably accurate association of color for each wavelength. Notice that the peak energy (top curve) of the Sun is found at a wavelength of around 450 nm (Wiehrli ’85 data set is 450.5nm and Thuilier 2002 is 456.6nm). This peak is in the blue portion of the spectrum. Does this surprise you? Did you think it was yellow? But, this doesn’t mean the Sun is a blue star, however. Unfortunately, the blue peak and the narrow band of yellow are only pieces of the puzzle. There are other color phenomena that must be considered.
Metamers also can contribute to a color determination and must be mentioned. We can produce one color (hue) by simply using two or three different colors. That’s how color is produced on tv’s and monitors. You know that, right? [Some green birds are not actually green but are blue, due to scattering effects, combined with yellow, which causes us to see green.] So, if we combine green light with orange and red, we might get yellow color result.
Does this color combination give us a yellow Sun? Well, that’s a tough question, but rather than tackle it head-on you’ll be happy to learn we can dodge it instead, thankfully. Take a look at the second, lower line in the graph: the “Photon flux”.
Our eyes, as well as, electronic sensors, are better represented as seeing photons rather than wavelengths of light. Since E = h f (photon energy = Planck’s constant times the frequency of the photon), then we can convert our spectral irradiance to a photon flux distribution. Notice how flat the distribution is. It is surprisingly flat. By sheer luck or madness, the peak photon flux just happens to be in the narrow yellow portion of the spectrum. I have seen several scientists claim the Sun is yellow because the peak wavelength of the Sun is in the yellow. However, there is no real peak; there is only a pimple, and a small one at that. One tiny pimple does not a complexion make, or destroy.
What color do you think you would get if you have such a flat photon flux distribution? Isn’t white the color we are suppose to see? Well……you’d think so, but if it is, it isn’t a definition I’ve been able to find, at least not one that is backed-up with objective evidence. Yet, admittedly, it does seem that a white appearance would be a logical result.
One problem with this idea comes from the sensitivity of our color cones. Will a flat photon flux distribution of light produce a "flat" signal to our brain? Nope, the combined spectral sensitivity of our color cones allows us to see green much more easily than any other color. The color we finally “see” in our mind is determined by a product of the spectral irradiance of the object as it enters our eye and the spectral sensitivity of our eyes (ignoring reflection properties if we are observing an illuminated object). [Many emergency vehicles are now bright green for this reason; it’s the last color visible as light dims. Red becomes black too quickly in dim light.]
So, oh great heliochromologist, you ask, where does all this take us and why not just tell us the answer? Well, this is a quest, not a fast food approachwe could take advantage of computer models that produce color results given the spectral irradiance, or we could use a number of other models that are used in industry and in association with CIE standards. I did find one impressive computer model effort done to determine the Sun’s color and their result is... peachy pink! Yikes, it’s a girl star! [Ok, that’s a little rude on my part, and, in fairness, they actually stated it to be pinkish peach, but remember, I’m in this for fun; Fun in the Sun.]
Fortunately, we also know the spectral irradiance of the Sun as observed terrestrially. The difference between an AMO and, say, an AM1 allows us to see just how much color is taken away (extinctions) by our atmosphere. Due to Rayleigh scattering, the primary color loss is in the blue portion of the spectrum. This is a very key point to the Sun’s color determination, because we must take into consideration this light loss caused by our atmosphere to all of our terrestrial observations.
But the final result comes from what has been discussed and actual observations. The evidence that produces strong support for the color conjecture for the Sun comes from the following separate lines of evidence:
1) Color images of several Solar twins taken from Kitt Peak. Especially, 18 Sco.
2) The study of cloud colors and other objects, adjusted for atmospheric effects.
3) Color images of the Sun from Kitt Peak.
4) Spectral irradiance data
5) Results from a new invention for heliochromology called a asterochromograph. [More than a colorimeter as it produces an actual true color spot by reproducing the spectral irradiance of any object.]
These observational inquiries will be discussed and the final color result will be given in the next and last blog in this...Quest for the Color of the Sun.






Comments