Whenever a scientist gives a public presentation related to energy, he or she provides some shocking facts that serve to motivate the research, but—ironically—depress the audience. When it comes to solar energy, my favorite is this: more energy strikes the surface of the earth in the form of sunlight in an hour than the entire human population consumes in a year. Solar energy, however, is dilute, and thus must be collected over a large area. Chemists Nathan Lewis and Daniel Nocera remarked that for solar energy to compete with fossil fuels in the United States, solar panels would have to cost, at most, ten dollars per square meter (or ten times the cost of paint) and would have to cover about one percent of the contiguous U.S.[1] This amount of land is comparable to the nation’s highway system and would cost about twenty trillion dollars.[2] The obscene cost of conventional solar cells is why the large-scale use of solar power is regarded as a pipe dream.
The most expensive part of conventional solar cells is the material of which they are made, polysilicon. Polysilicon comes from sand in a multi-step process that is extraordinarily energy-intensive. A mere kilogram costs about a gigajoule of energy, which is the amount of hydroelectricity the Hoover Dam produces in one second.[3] Arguably, the only photovoltaic materials that are remotely similar to paint—in terms of both price and the ease with which they can be coated on surfaces—are organic semiconductors.
Organic semiconductors are soft, carbon-based materials synthesized from products (often byproducts) of the petroleum industry. The molecular structures of these materials have an alternating arrangement of single and double bonds between carbon atoms. A system of atoms arranged in this way is said to be conjugated; conjugation is the basis of everyday effects such as the colors of chlorophyll and beta-carotene. Such a molecule accommodates electric charge relatively easily, by allowing the charge to spread out over all its atoms. Conjugated molecules together form a solid semiconductor, with properties like those of silicon.
When light strikes an organic semiconductor, it knocks an electron loose within the material. A positive charge, called a hole, is left behind where the electron used to be. Newly formed electron-hole pairs can travel within the material about ten nanometers—the diffusion length—before they collapse, or recombine into nothingness. Some organic semiconductors are better at carrying electrons (“n-type”); others are better at carrying holes (“p-type”).[4] Both types of charges must split from each other at an interface between n- and p-type materials before they can go their separate ways to power devices.
The absorption of light and the separation of charges take place within a thin film of about one hundred nanometers. This film contains both n- and p-type materials, and is sandwiched between two types of electrodes: a transparent electrode and a reflective electrode. The transparent electrode usually collects holes, while the reflective electrode usually collects electrons.
Figure “a” is a drawing of the simplest type of plastic solar cell: the “sandwich-type solar cell”, or planar heterojunction. The problem with this geometry is that an electron-hole pair can only travel about ten nanometers before it collapses,[5] while the thickness the material requires to absorb most of the incoming light is about a hundred nanometers. So, most electron-hole pairs are wasted: they simply collapse before reaching the junction at which they can split into free charges. The best sandwich-type plastic solar cells are around 1% efficient.[6]
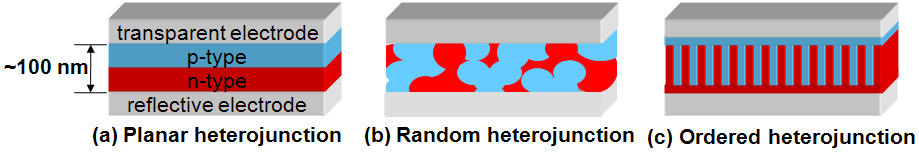 The state of the art in plastic solar cells has an intermixed layer—a random heterojunction—between the n-type and p-type materials (Figure “b”). The random heterojunction is like a molecular version of spaghetti and meatballs. It provides a large area for splitting electron-hole pairs, but contains many isolated blobs of n- and p-type materials, which are dead ends for electrons and holes. Efficiencies reported for random heterojunctions have hovered around 5% since 2004, while the number of papers published per year on this type of device will have increased by four times in 2009.[7]
The state of the art in plastic solar cells has an intermixed layer—a random heterojunction—between the n-type and p-type materials (Figure “b”). The random heterojunction is like a molecular version of spaghetti and meatballs. It provides a large area for splitting electron-hole pairs, but contains many isolated blobs of n- and p-type materials, which are dead ends for electrons and holes. Efficiencies reported for random heterojunctions have hovered around 5% since 2004, while the number of papers published per year on this type of device will have increased by four times in 2009.[7]Rather than developing—from the bottom up—new materials and manufacturing processes that yield devices with superior properties, research has focused on tweaking top-down parameters that are easy to implement (such as heating temperatures and processing solvents), but are unlikely to improve the devices in any fundamental way. It seems improbable that the random heterojunction will reach the goal of 10%, given this deceleration in progress.
One of the strategies that could increase the mobility of electron-hole pairs is to design materials whose molecules pack together efficiently in a solid. Crystalline packing generally augments the mobility of charges in organic semiconductors. Another strategy is to choose a pair of n- and p-type materials that undergo fluorescent resonant energy transfer, which is a way of transferring energy between molecules over distances much larger than electron-hole pairs can diffuse on their own.[8] Theoretical modeling will aid in the design of new materials, whose chemical structures must be simple enough for chemists to synthesize inexpensively on the megaton scale.
We might, however, be stuck with materials with poor diffusion properties, in which case we have to design a device that incorporates the best features of the planar and bulk heterojunction. Imagine n- and p-type materials that fit together like two large Lego plates, rather than like spaghetti and meatballs. A cross section of this ordered heterojunction would look like interdigitated fingers where the lateral width of each finger was equal to the diffusion length, while the thickness was that at which light absorption was optimal (Figure “c”).[9] One way to make this structure would be to mold the n-type material with trenches, and then fill in the gaps with the p-type material. A new technology to mold materials with such fine features must be developed to make such a solar cell on a large scale. (CDs, DVDs, and Blu-ray Discs are manufactured by molding, but the imprinted features are nearly two orders of magnitude too big for solar cells.)
If efficient plastic solar cells could be made inexpensively, what would be the outcome? The dream is that energy would be revolutionized through the use of cheap solar material that could be paved onto flat expanses or spray-painted onto cars. Realistic and short-term applications for plastic solar cells include lightweight power sources for portable (even wearable) electronics; low-cost, off-grid power for rural areas of developing countries; and other applications in which low cost, rather than state-of-the-art efficiency, is the goal.
It is ironic that plastic solar cells could provide an alternative to the combustion of fossil fuels, but are dependent on the petroleum industry for starting materials. Plastic solar cells could thus become a lucrative—but unexpected—cross-product between alternative energy research and oil-rich countries. The King Abdullah University of Science and Technology in Saudi Arabia has initiated an international collaboration on organic photovoltaics with both academic and industrial contributors.[10] Saudi Arabia’s interest in solar power at first seems contradictory to its dependence on revenue from oil, but the reason is quite logical. That is, the Arabian Peninsula has both of the natural resources for large-scale manufacture and use of plastic solar cells: abundant petroleum and sunlight. Many other regions (e.g., South America and Texas) share these qualities.
Plastic solar cells could become an important source of renewable energy, but more research is required to overcome limitations in efficiency.[11] One of the most exciting aspects of this research is that it is interdisciplinary: it is equal parts chemistry, physics, materials science, and engineering; even biology is likely to play a role as the molecular mechanisms of photosynthesis become applicable to devices. A successful outcome for the field will require new ideas, hard work, generous funding, and luck.
References
[1] N. S. Lewis, D. G. Nocera, “Powering the Planet: Chemical Challenges in Solar Energy Utilization” Proceedings of the National Academy of Sciences of the USA (2006) Vol. 103, p. 15729. See videos available at http://nsl.caltech.edu/energy.html.
[2] This is a back-of-the-envelope calculation. One percent of the contiguous U.S. is 80 billion square meters; the cost of conventional solar panels is about 300 dollars per square meter.
[3] E. D. Williams, R. U. Ayers, M. Heller, "The 1.7 Kilogram Microchip: Energy and Material Use in the Production of Semiconductor Devices" Environmental Science and Technology (2002) Vol. 36, p. 5504.
[4] The nomenclature of n- and p-type for organics is different from its use for conventional semiconductors. It refers to the type of charge injected into the material based on the proximity of the molecular orbitals to the Fermi energies of typical electrodes, and other extrinsic effects. See V. Coropceanu, J. Cornil, D. A. da Silva Filho, Y. Olivier, R. Silbey, J.-L. Bredas, “Charge Transport in Organic Semiconductors” Chemical Reviews (2007) Vol. 107, p. 926.
[5] In a conventional solar cell, free charges carriers are generated directly upon the absorption of light. Electron-hole pairs are tightly bound in organic materials because of the low dielectric constant.
[6] M. M. Alam, S. A. Jenekhe, “Efficient Solar Cells from Layered Nanostructures of Donor and Acceptor Conjugated Polymers” Chemistry of Materials (2004) Vol. 16, p. 4647.
[7] A literature search on ISI Web of Science for “bulk heterojunction” yielded 77 hits for all of 2004 and 233 from January through August of 2009.
[8] S. R. Scully, P. B. Armstrong, C. Edder, J. M. J. Frechet, M. D. McGehee, “Long-Range Resonant Energy Transfer for Enhanced Exciton Harvesting for Organic Solar Cells” Advanced Materials (2007) Vol. 19, p. 2961.
[9] S. Gunes, H. Neugebauer N. S. Sariciftci, “Conjugated Polymer-Based Organic Solar Cells” Chemical Reviews (2007) Vol. 107, p. 1324.
[10] M. D. McGehee “Molecular Photovoltaics as a Low-Cost Renewable Energy Source” downloaded from http://www.kaust.edu.sa/research/pdf/mcgehee.pdf on 21 September 2009.
[11] Solar energy could only be a primary source of energy if it could be stored for use at night. One elegant strategy would be to store the energy in the form of chemical bonds. See M. W. Kanan and D. G. Nocera “In Situ Formation of an Oxygen-Evolving Catalyst in Neutral Water Containing Phosphate and Co2+” Science (2008) Vol. 321, p. 1072.





