A closer look at one lineage, or clone, of a D. discoideum farmer carries not one but two strains of bacteria. One strain is the “seed corn” for a crop of edible bacteria, and the other strain is a weapon that produces defensive chemicals. The edible bacteria, the scientists found, evolved from the toxic one. The two strains differ by many mutations but a single key mutation, which hit an important controller in the genome of the nonfood strain, alters expression of 10 percent of its genome. This alteration increases the expression of some genes and decreases the expression of others.
A mutation that affects this much of a genome could be lethal, but in this case it had the surprising effect of making the bacterium edible by changing its chemical profile.
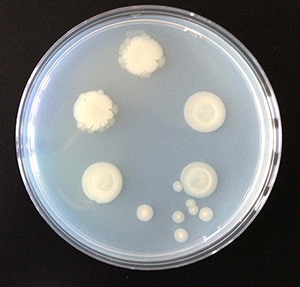
Colony morphology when bacteria carried by one D. discoideum farmer clone were cultured. Brock noticed some of the bacterial colonies had fuzzy borders and others had smooth ones. Credit: Dr. Debra Brock
When researcher and new paper co-author Dr. Debra Brock compared the standard axenic (pure, or uncontaminated) lab clone of D. discoideum with D. discoideum collected from the wild, she saw bacteria in the sori of some clones. Oddly it was always the same clones that carried bacteria. The bacteria caught her attention because she had never seen anything like this in the lab clone.
Whenever she found a D. discoideum clone carrying bacteria, Brock tried to isolate the bacteria. This was a bit hit or miss because many organisms that live in the soil cannot be grown in the lab. Eventually she found a champion D. discoideum: a farmer clone from which she was able to isolate two strains of bacteria. At least the strains looked different when they were cultured in a dish. She sent the two bacteria out to be identified genetically and both came back as Pseudomonas fluorescens: the same species, even though they were morphologically so different.
“It was a bit of a puzzle,” Brock said. On top of that one of the two morphs was edible and the other was not, and the edible one was the first edible strain she had isolated that wasn’t a lab feedstock. “So, I now had two bacteria that seemed the same and one was a food and the other wasn’t. That was really odd.”
Guns and butter...
The Washington University of St. Louis researchers sent the bacteria to Prof. Jon Clardy's lab at Harvard, where Dr. Pierre Stallforth, a post-doctoral associate in the Clardy lab, grew them in liquid media. He sent extracts from the media back to Brock, who tested them on D. discoideum to see if they were active.
“Ultimately Pierre figured out that the nonfood strain was producing two chemicals: chromene and pyrrolnitrin. And excitingly, chromene is a new compound,” said co-author Professor Joan Strassmann. “We determined chromene increases spore production in the farmer strain and suppresses spore formation in the nonfarmer strain. We saw the same increases in the farmer and decreases in the non-farmer with pyrrolnitrin. A known antibiotic and antifungal, pyrrolnitrin probably also suppresses other organisms in the soil that might compete with the farmer strain.”
Assays showed that it was not merely the absence of chromene and pyrrolnitrin that made the food bacterium edible. Something else is going on as well.
...but why become butter?
Stallforth next sequenced the entire genome of the two bacterial strains to look for mutations that might explain the differences between them. The genes responsible for producing pyrrolnitrin were intact in both strains. So he looked at the genes for a two-part global activator that regulates the pyrrolnitrin pathway, among many other genes.
Sure enough, there was a mutation in one of the controller genes of the food bacterium that turned it off and broke the controller. As shown by others in a previous study, breaking the controller changed the expression of 10 percent of the bacteria’s genome.
“That’s pretty cool, but then you still don’t really know for sure if that mutation is the one that matters,” Strassmann said.
To check, Stallforth artificially broke the controller — and only the controller — in the nonedible P. fluorescens bacterium. The knockout strain he created had the same chemical profile as the food bacterium and it, too, was edible.
Had a similar mutation in the evolutionary past created the edible strain? To answer that question, the scientists constructed a family tree of P. fluorescens clones in the Strassmann/Queller lab by comparing 20 genes.
“It turns out that of all the bacteria strains we’ve ever isolated, the two we collected from the D. discoideum farmer clone Brock discovered are the most closely related," said co-author Professor David Queller. “The tree also tells us that edibility is a derived trait. These guys used to be inedible and became edible. That’s just a weird thing to evolve: to be able to eaten."
It makes sense only because it benefits kin, more of whom will be carried to new locations by the well-fed farmer D. discoideum clone, the scientists said.
It’s altruism, ultimately. Altruism in miniature.
This isn't the first time D. discoideum has been found altruistic. Prior studies here showed how they were loners in times of plenty but got more 'altruistic' in resource-scarce situations.
Citation: Pierre Stallforth, Debra A. Brock, Alexandra M. Cantley, Xiangjun Tian, David C. Queller, Joan E. Strassmann, and Jon Clardy, 'A bacterial symbiont is converted from an inedible producer of beneficial molecules into food by a single mutation in the gacA gene', PNAS July 29, 2013, doi:10.1073/pnas.1308199110






Comments