A medicated silicone ring that rests on the surface of the eye reduced eye pressure in glaucoma patients by about 20 percent over six months, potentially benefiting 3 million people in the United States who have glaucoma. Phase 2 clinical trial results on this technology were published today and the results are also being presented today at the Ophthalmology Innovation Summit in New Orleans.
When internal eye pressure becomes too high, it can damage the optic nerve and cause vision loss. Researchers found that a silicone ring treated with the glaucoma medication bimatoprost lowered internal eye pressure in patients with glaucoma or ocular hypertension by about 20 percent over six months. It's also a non-invasive treatment: the ring rests on top of the eye underneath the lids and does not require surgery to install. Some patients experienced itching and eye redness, which are not unusual when taking glaucoma treatment drops.
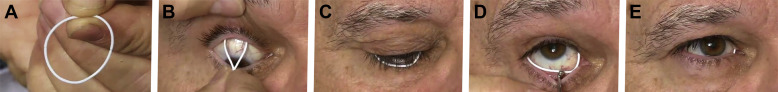
Photographs showing the method of placement of a bimatoprost ocular insert. A, The upper lid is retracted manually and the insert is placed in upper fornix by the physician. After (B) placement of the top half of the insert in the upper fornix, (C) the lower lid margin is retracted gently either manually or with a scleral depressor (D) to seat the bottom half of insert into the lower fornix. E, Insert in situ with a small portion of the insert visible in the medial canthus. Credit: DOI: 10.1016/j.ophtha.2016.04.026.
Authors said the device was well-tolerated and safe, with a high retention rate of 89 percent for both groups at six months. The ring became dislodged in 15 patients but was replaced each time, allowing therapy to continue. Some patients experienced itchiness and eye redness, which is not unusual for patients taking glaucoma medication.
“In making effective treatments easier for patients, the hope is that we can reduce vision loss from glaucoma, and possibly other diseases,” said study author James D. Brandt, M.D., director of the UC Davis Medical Center Glaucoma Service. “What is exciting is that this is just one of several sustained-release drug delivery methods designed to help patients who have trouble taking daily eye drops.”
A phase 3 study of a larger group of patients is expected to begin later in 2016.
“Six-month IOP Reduction with a Topical Bimatoprost Ocular Insert: Results of a Phase 2 Randomized Controlled Study,” Brandt, et al. Ophthalmology, article in press, May 2016. DOI: 10.1016/j.ophtha.2016.04.026.






Comments