It wasn't a linear path but after a lot of trial and error, and some fitful starts (including deaths, the kind of thing that would get a product pulled from existence in today's cancel culture, the Bacillus Calmette–Guérin (BCG) vaccine has been administered routinely to protect babies against tuberculosis since 1921.
Today only a few countries, such as the United States and the Holland (where TB is rare) don't use it.
But it is given to infants via needle and does well against disseminated TB but is less effective at preventing pulmonary TB, the major cause of illness and deaths, in teens or adults.
A new study in rhesus macaques shows doctors can change all that.
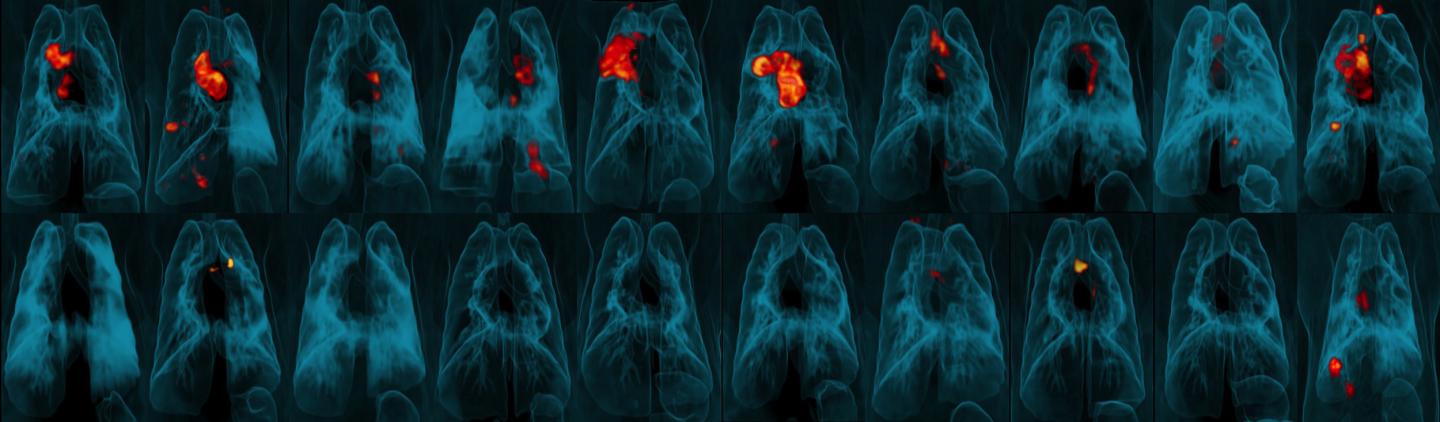
3-D PET-CT scans of lungs showing areas of TB infection and tissue inflammation (red and orange) in macaques challenged with Mtb after vaccination with either ID BCG (top row) or IV BCG (bottom). Credit: University of Pittsburgh School of Medicine
To control Mtb infection and prevent clinical disease, a TB vaccine must elicit strong, sustained responses from the immune system's T cells, specifically those in the lungs. However, the standard, ID, route of BCG administration may not generate enough of these critical cells in the lungs. The NIAID researchers and their colleagues hypothesized that administration of BCG by IV or aerosol (AE) routes would overcome this hurdle and thus confer substantially better protection from infection and/or disease in rhesus macaques following challenge with virulent Mtb.
In their study, groups of animals received the BGC vaccine by ID, AE or IV routes. The scientists assessed immune responses in blood and in fluid drawn from the lungs for a 24-week period following vaccination. IV BCG vaccination resulted in the highest durable levels of T cells in the blood and lungs.
Six months after vaccination, the researchers exposed groups of vaccinated rhesus macaques (immunized via ID, AE or IV routes) and a group of unvaccinated macaques to a virulent strain of Mtb by introducing the bacteria directly into the animals' lungs. They then tracked the infection and disease development over three months. Nine out of 10 animals vaccinated with IV BCG were highly protected; six showed no detectable infection in any tissue tested and three had only very low counts of Mtb bacteria in lung tissue. All unvaccinated animals and those immunized via ID or AE routes showed signs of significantly greater infection.
The investigators concluded that IV BCG conferred an unprecedented degree of protection in an animal model of severe TB and "represents a major step forward in the field of TB vaccine research."






Comments