So say US Peroxide, a company marketing ‘Technologies for a Clean Environment’.
Today, Fenton's Reagent is used to treat a variety of industrial wastes containing a range of toxic organic compounds (phenols, formaldehyde, BTEX, and complex wastes derived from dyestuffs, pesticides, wood preservatives, plastics additives, and rubber chemicals). The process may be applied to wastewaters, sludges, or contaminated soils, with the effects being:
* Organic pollutant destruction
* Toxicity reduction
* Biodegradability improvement
* Biological / Chemical Oxygen Demand removal
* Odor and color removal
Now I am not an agent of theirs, although I do approve of hydrogen peroxide as an environmentally friendly cleaner and disinfectant. Unlike chlorine containing bleaches, it does not release chloride ions which accelerate the corrosion of metal fittings on one’s ‘convenience’.
However, I quote them as they help me in writing this blog. I was searching for Fenton’s reagent after seeing this on the Science Codex:
See off Alzheimer's with the color purple
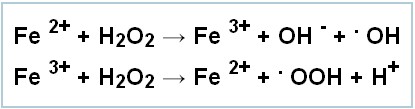
Ground-breaking research from Professor Douglas Kell, published in the journal Archives of Toxicology, has found that the majority of debilitating illnesses are in part caused by poorly-bound iron which causes the production of dangerous toxins that can react with the components of living systems.It was this that reminded me of Fenton’s reagent, which produces hydroxyl radicals by the following reaction:
These toxins, called hydroxyl radicals, cause degenerative diseases of many kinds in different parts of the body.
Picture courtesy of US Peroxide again. In their chemistry page, they give this table:
Reactive Species Relative Oxidation Power (Cl2=1.0)
Fluorine 2.23
Hydroxyl radical 2.06
Atomic oxygen (singlet) 1.78
Hydrogen peroxide 1.31
Perhydroxyl radical 1.25
Permanganate 1.24
Hypobromous acid 1.17
Chlorine dioxide 1.15
Hypochlorous acid 1.1
Hypiodous acid 1.07
Chlorine 1
Bromine 0.8
Iodine 0.54
That seems to put Fenton’s reagent right up next to fluorine, which is a nightmare to contain and handle. However, this is thermodynamic oxidation power, not reactivity, so your Fenton’s will sit there nicely in a test-tube and not jump out at you.
The research review suggests that the pigments of purple fruit are just the thing to catch hold of that chelated iron and stop it reacting with peroxide naturally present in the body. A chemical species which is powerful enough to clean up waste water circulating among one’s tissues? The thought of hydroxyl running wild among one’s neurons and leading to Alzheimer’s is enough to send one rushing to the nearest greengrocer.
However, the UK NetDoctor now tells us
Purple fruit health claims premature
saying that the case is far from proven, and that
Crucially, though, there is still a need for more proof that food can have a meaningful effect on these specific pathways in humans. It would appear that the next steps in the exploration of this theory would be to establish the action of the individual compounds found in foods such as purple fruit and to test whether candidate foods affect either health outcomes or iron chelation in humans. Such research is likely to be long and complex.Returning to the main article, you can link to this paper here, and it seems that Springer are giving you free access till the end of this year.
However, even if the compounds in purple berries are just the ticket, how would they get from the alimentary canal into the bloodstream and then cross the blood-brain barrier?
Or is it, I wonder, that the chelation must happen in the gut, before the iron gets into the bloodstream?





Comments