
I love separation science, since it amuses me no end. As the coffee stain still lurks at my desk, reading through this article, the stain will be a mainstay at my laboratory. Okay, the hygiene issues will linger. Scientists at Harvard, California and Stanford universities have come up with use of coffee ring effect. A chromatography method that uses the same physics as the coffee stain: It separates nanometer- and micrometer-scale particles by size as a droplet dries.
Considering that coffee is a colloidal suspension, when coffee droplets dry on a clean surface, the evaporating liquid pushes the particles to the edge, concentrating them and leaving a ring at the droplet rim.
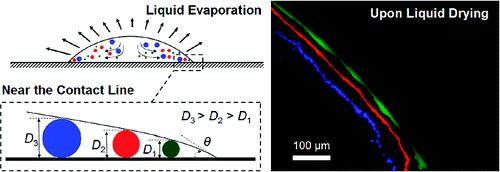
All this while, we may consider that the particles just concentrate at the rim of the coffee stain, they are working on theory as to how the particles move as the droplet evaporates. Here comes in the theory of particle separating capacity. As the droplet grew thinner towards its edge, the particles would stop moving at a point where their diameter matched the droplet's height.
Courtesy: Analytical Chemistry
They elucidated the theory through an experiment wherein they made an aqueous suspension of fluorescent beads with diameters of 40 nm, 1 μm, and 2 μm. They let a 0.5-μL droplet of the suspension dry at room temperature. Three distinct rings appeared, with the smallest beads in the outermost ring and the largest ones in the innermost ring. The technique is claimed to separate macromolecules, proteins, bacteria, and mammalian cells in an aqueous mixture, that too without any use of external power source.
This technique definitely works well as I tried to figure out the separation of some amino acids using this theory. Well, this makes sense especially in a country like India where resources are scarce. Though, this may raise some hygiene issues, if I allow that stain to remain on my table, but at this point of time I need some coffee stain.
Coffee Stains…… Anyone !





Comments