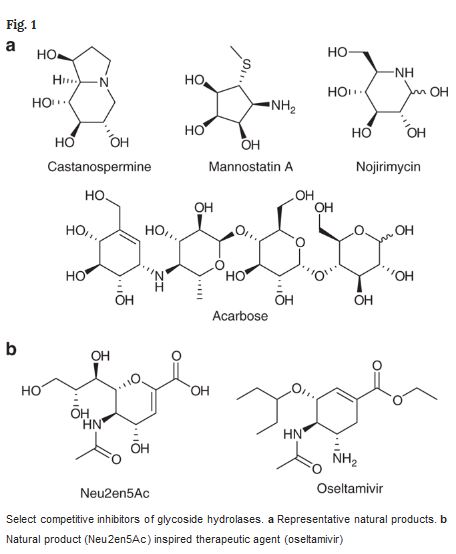
Glycosides are essential enzymes to digest carbohydrates but they are also key players in infections caused by pathogens, in anti-bacterial defense and many other vital cellular processes. Because these small molecules that are able to bond with and inhibit the activity of enzymes in infectious diseases, it opens up the basis for new medicines.
“We have synthesized new molecules, taken new measurements of their inhibitory activity, obtained structures by way of x-ray diffraction and then conducted computer simulations on the whole process,” says Vicent Moliner, the head of the Computer Biochemistry group at Universitat Jaume I in Castellón, Spain. He notes the data obtained through computer simulations coincide with the experimental data of their colleagues in Canada and the United Kingdom and make it possible to explain how these inhibitors bond with the enzyme.
Citation: W. Ren, R. Pengelly, M. Farren-Dai, S. Shamsi Kazem Abadi, V. Oehler, O. Akintola, J. Draper, M. Meanwell, S. Chakladar, K. Świderek, V. Moliner, R. Britton, T. M. Gloster, A. J. Bennet: "Revealing the mechanism for covalent inhibition of glycoside hydrolases by carbasugars at an atomic level". Nature Communication 9, Article number: 3243 (2018). DOI: 10.1038/s41467-018-05702-7






Comments