But there has long been another possibility; a kind of “oxygen-free respiration” hinted at in studies on primordial microbes that are still found in anoxic habitats today. There have been found genes in microbes that perhaps encode for a primordial respiration enzyme.
Yet proving the existence of this respiratory enzyme and isolating it requires time and effort. The complex is fragile and often fell apart during attempts to isolate it from the membrane.
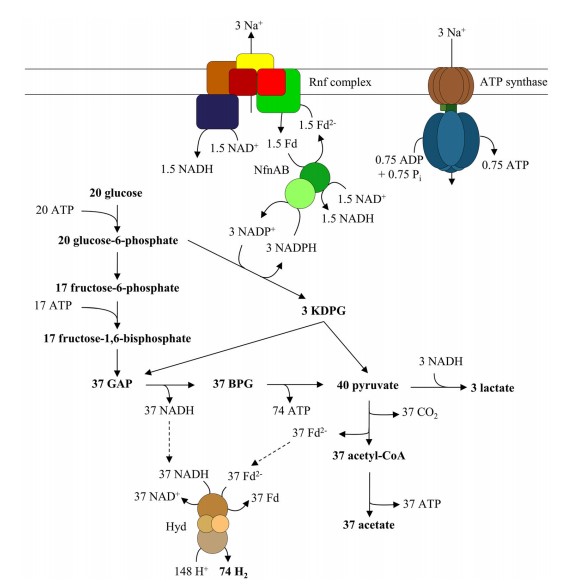
Glucose is oxidized via glycolysis and Entner-Doudoroff pathway and acetate is produced. NADPH generated in the glucose-6-phosphate dehydrogenase reaction is oxidized with an NADH-dependent Fd2−:NADP+-transhydrogenase (Nfn) by reducing Fd and NAD. Electrons from Fd2− are shuffled to NAD by the Rnf complex, thereby generating an Na+ gradient that is used by F1FO ATP synthase to generate ATP. NADH generated by the Rnf complex and Nfn is used to reduce pyruvate to lactate. All other reducing equivalents are used by the electronbifurcating/confurcating hydrogenase (Hyd) to generate H2. KDPG 2-keto-3-desoxy-6-phosphogluconate, GAP glyceraldehyde 3-phosphate, BPG bisphosphoglycerate. Note that the ion/electron and ion/ATP stoichiometries are based on thermodynamics
A team at Goethe University used a heat-loving bacterium, Thermotoga maritima, which grows at temperatures between 60 and 90°C and also contains Rnf genes, to see if the Rnf enzyme in this bacterium would be a bit more stable. Their success led to a recent paper. They report that the enzyme complex functions a bit like a pumped-storage power plant that pumps water into a lake higher up and produces electricity via a turbine from the water flowing back down again.
In the bacterial cell does the ferredoxin:NAD-oxidoreductase Rnf enzyme transports sodium ions out of the cell’s interior via the cell membrane to the outside and in so doing produces an electric field. This electric field is used to drive a cellular “turbine” (ATP synthase): It allows the sodium ions to flow back along the electric field into the cell’s interior and in so doing it obtains energy in the form of the cellular energy currency ATP - like we humans use.
The biochemical proof and the bioenergetic characterization of this primordial Rnf enzyme explains how first forms of life produced the central energy currency ATP. The Rnf enzyme evidently functions so well that it is still contained in many bacteria and some archaea today, in some pathogenic bacteria as well where the role of the Rnf enzyme is still entirely unclear.
Citation: Kuhns, M, Trifunovic, D., Huber, H., Müller, V. (2020). The Rnf complex is a Na+ coupled respiratory enzyme in a fermenting bacterium, Thermotoga maritima. Communications Biology, DOI 10.1038/s42003-020-01158-y






Comments