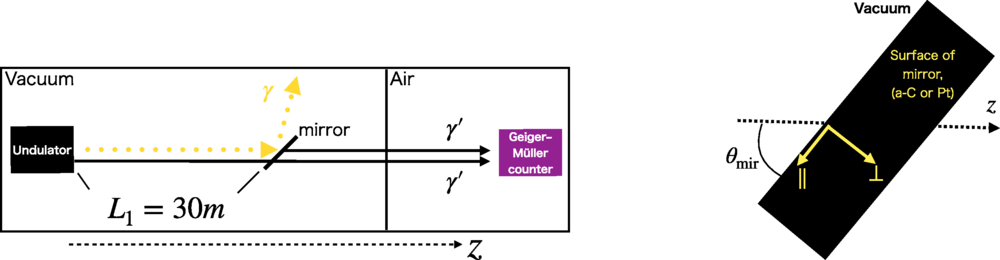
 Synchrotron Could Shed Light On Exotic Dark Photons
Synchrotron Could Shed Light On Exotic Dark PhotonsThere are many hypothetical particles proposed to explain dark matter and one idea to explore how...
 The Pain Scale Is Broken But This May Fix It
The Pain Scale Is Broken But This May Fix ItChronic pain is reported by over 20 percent of the global population but there is no scientific...
 Study Links Antidepressants, Beta-blockers and Statins To Increased Autism Risk
Study Links Antidepressants, Beta-blockers and Statins To Increased Autism RiskAn analysis of 6.14 million maternal-child health records has linked prescription medications...
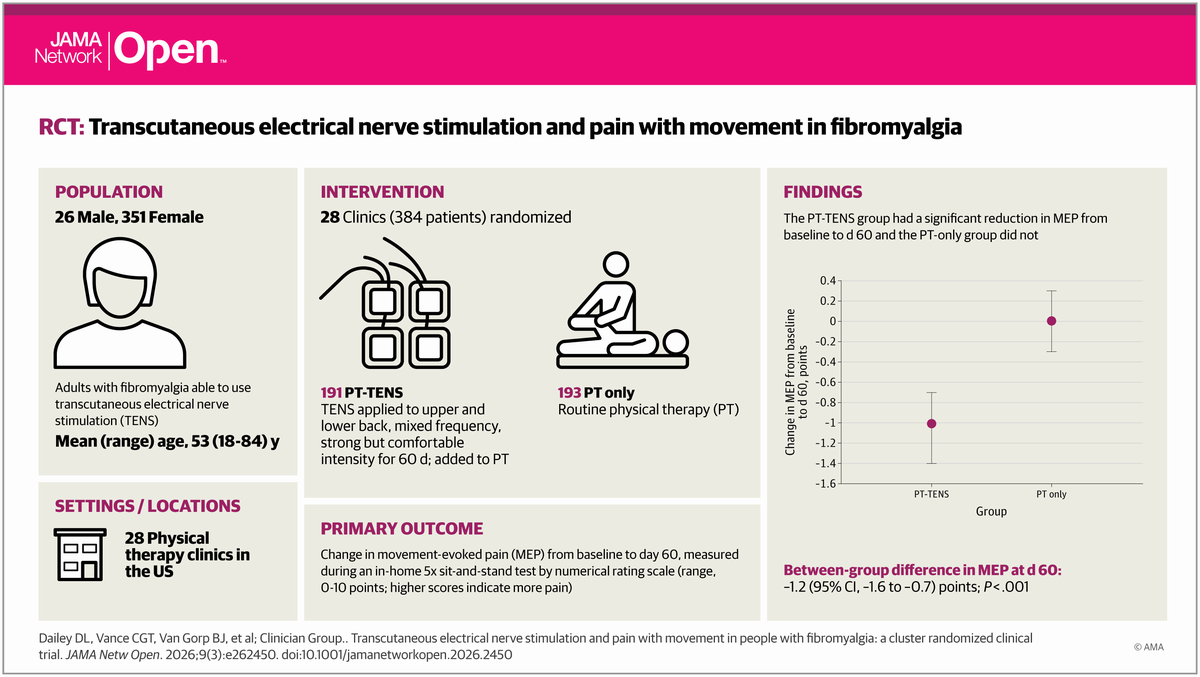 Pilot Study: Fibromyalgia Fatigue Improved By TENS Therapy
Pilot Study: Fibromyalgia Fatigue Improved By TENS TherapyFibromyalgia is the term for a poorly-understood condition where people experience pain and fatigue...