A team of scientists has provided, for the first time, a detailed map of how the building blocks of chromosomes, the cellular structures that contain genes, are organized in the fruit fly Drosophila melanogaster. The work identifies a critical stop sign for transcription, the first step in gene expression, and has implications for understanding how the AIDS virus regulates its genes.
The scientists found that nucleosomes--chromosomal building blocks made up of proteins around which DNA is coiled--occur at precise locations along genes that are actively undergoing transcription. They also showed that RNA polymerase--the enzyme that reads genes as the first step in making proteins--is stopped at the first nucleosome, where it remains idle until it is directed to continue moving forward.
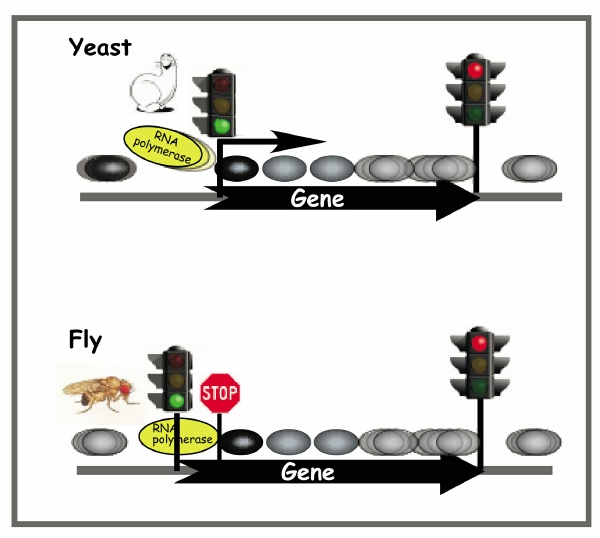
"This discovery is important because nucleosomes are barriers to transcription and we now are seeing the impact of nucleosome organization on RNA polymerase," said lead investigator B. Franklin Pugh, professor and Willaman Chair in Molecular Biology at Penn State University.
Using state-of-the-art ChIP-sequencing, a genome-mapping tool provided by collaborator Stephen S. Schuster, Penn State professor of biochemistry and molecular biology, and computational predictions developed by collaborators Ilya Ioshikhes, an assistant professor at Ohio State University, and Istvan Albert, a research assistant professor of bioinformatics at Penn State, the scientists precisely mapped the locations of hundreds of thousands of nucleosomes. The scientists then compared these maps to the team's earlier maps of the baker's yeast Saccharomyces cerevisiae, revealing that evolution has organized nucleosomes differently in simple life forms compared to more complex organisms like the fruit fly.
In yeast, a nucleosome sits on top of the transcription start site, so RNA polymerase must contend with that nucleosome as soon as it begins to transcribe the gene. In contrast, nucleosomes are positioned further downstream in fruit flies, so transcription starts but then soon pauses at the first nucleosome the RNA polymerase encounters. "This pause is maintained until chemical signals from the cell cue the removal of the nucleosome and encourage the RNA polymerase to continue along its path," said key collaborator David S. Gilmour, professor of molecular and cellular biology at Penn State and an expert on the pausing of RNA polymerase.
"A year ago, we could name about 10 genes that work this way. Now, we know of 1,000 in flies alone and we suspect there could be many more in humans," said Gilmour. "Even HIV genes have a paused RNA polymerase. Release of this pause may be key to activating HIV replication of otherwise latent viruses. Taking advantage of this new understanding might enable the development of more effective anti-viral drugs," he said.
"The bottom line is that we need to know how the expression of genes is regulated in order to understand the underpinnings of most human diseases, and these findings take us one step closer," said Pugh.
This work was funded by the National Institutes of Health and Penn State. The findings will be published in the 15 May 2008 issue of the journal Nature.






Comments