Using a combination of genetic and optical techniques, researchers at the Champalimaud Neuroscience Programme have established the effect of serotonin on sensitivity to pain.
"Serotonin is a small molecule known to be implicated in a wide range of brain functions, from the control of sleep and appetite, to the regulation of complex emotional behaviours, This neurotransmitter is also popularly thought to contribute to feelings of well being and happiness, as some anti-depression medications work through increasing serotonin in the brain," says Zachary Mainen, CNP director and principal investigator of the Systems Neuroscience Lab.
Serotonin's great importance led researchers to seek ways of understanding its function, but studying it has been a long-standing challenge.
"Most of the cells that produce serotonin are located in a defined cell group called the Dorsal Raphe Nucleus (DRN)," explains Zachary Mainen. "This cell group is small and located deep in the brain, which makes targeting it difficult. In addition, other cells that produce and release different molecules are also present in the DRN, which means that general stimulation of the area may result in the release of other molecules besides serotonin."
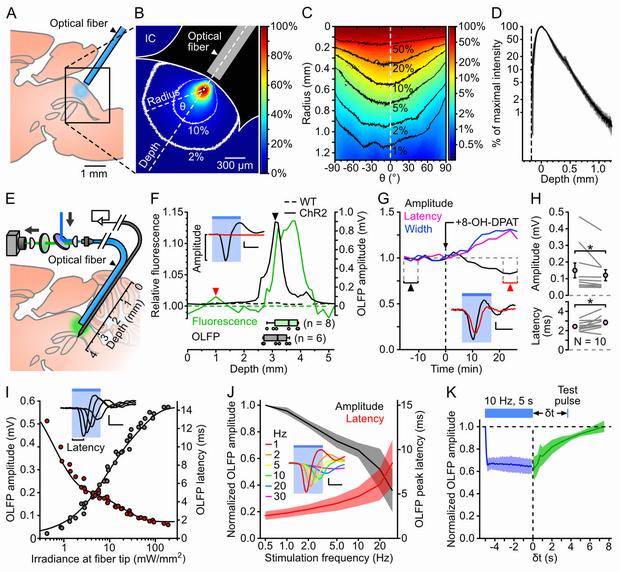
A, Schematics of the experiment used to assess the spread of blue light in the DRN.
B, Example image of DRN illumination. The intensity is color-coded relative to the pixel of maximal intensity (white dot). The 10 and 2% contour lines are shown in white. IC: inferior colliculus.
C, Circular profile plot calculated from B, showing pixel intensities along concentric lines centered on the brightest pixel.
D, Average intensity profile (± SD, shaded area; n = 3 brains) along the fiber axis. The depth and intensities are calculated relative to the brightest pixel.
E, Schematics of the setup used to map YFP fluorescence and photoevoked firing in vivo.
F, Example of a combined electrophysiological and fluorescence mapping experiment in a ChR2-YFP-expressing SERT-Cre (ChR2, solid lines) and a wild-type (WT, dotted lines) mouse. Top: normalized fluorescence (green, left axis) and OLFP amplitude (black, right axis) profiles plotted against the optrode position. Bottom: boxplots showing the optrode position at the point of maximal fluorescence (green, n = 8 mice) and largest OLFP amplitude (grey, n = 6 mice). Inset: average OLFP for the ChR2 mouse at the locations marked by the arrowheads.
G, Example showing the time course of the effect of the 5-HT1A receptor agonist 8-OH-DPAT on the OLFP amplitude (black), latency (purple) and width (blue). Bin: 158 s. Inset: average OLFP before (black) and after (red) 8-OH-DPAT injection (taken from the periods indicated by arrowheads).
H, Top: OLFP amplitude before and after 8-OH-DPAT injection (mean ± SD: 150±121 µV and 121±105 µV respectively, n = 10). Bottom: OLFP peak latency before and after 8-OH-DPAT injection (mean ± SD: 2.4±0.5 ms and 2.8±0.7 ms, n = 10). Changes are significant in both cases (P = 0.037 and P = 0.030 respectively, paired Wilcoxon rank sum test). Error bars represent the SEM.
I, Example illustrating the dependence of the OLFP amplitude (grey points) and latency (red points) on the irradiance at the fiber tip. The sum of two exponential functions was fitted to each curve (black lines). Inset: superimposed OLFPs for 0.2, 0.7, 1.6 and 6.0 mW·mm−2.
J, Normalized average (± SD, shaded area) OLFP amplitude (black, left axis) and latency (red, right axis) versus photostimulation frequency (n = 7 mice).
K, Average OLFP amplitude recovery curve (green; ± SD, shaded area) assessed by delivering test pulses at variable intervals (δt) after 10 Hz, 5 s trains of 6 ms light pulses (n = 4 mice). Blue: average amplitude during the trains.
doi:10.1371/journal.pone.0105941
"To overcome the limitations of previous studies and explore the specific function of serotonin, we used a combination of light and genetics, an approach called optogenetics," says Guillaume Dugué, a former postdoctoral researcher in the lab ofMainen.
Using genetic techniques, the researchers expressed a light-sensitive protein specifically in the serotonin-producing cells of mice, so that when the researchers shone light on these cells, the cells released serotonin.
"The effect of the serotonin was clear,"says Dugué. "Mice that we stimulated to release serotonin showed a significant decrease in sensitivity to pain, when compared with mice in the control group."
"We devoted substantial efforts to optimizing light activation of serotonin-producing cells. Overall these results provide a new level of evidence on the importance of serotonin in gating the influence of sensory inputs to behavioral outputs, a key physiological role that will help define large-scale theories of serotonin function. Moreover, it has possible implications for better understanding chronic pain treatment," concludes Mainen.






Comments