You see advertisements for fitness apps on smartphones all of the time. Apple prides itself on convincing you that you will be a better dancer and healthier if you buy their phone. The problem is that the people most likely to use a fitness app for more than a week are least likely to need it.
Or maybe they do, according to recent Scare Journalism. In the health fad culture perpetuated by mainstream media, there is now a War On Sitting. Once some crazy claim appears in the New York Times, studies are going to crop up affirming exactly what popular media claims say.
In America, we have gotten rid of most manufacturing jobs, many people instead work at a desk. That's good for global warming but bad for health. Epidemiologists, in their desire to find a statistical link to whatever is trending, associate sitting with a greater risk for obesity, cardiovascular disease and mortality. Miriam Hospital has created an app to try and get you off the couch, but to sound more serious they call it a smartphone-based intervention. It's in an open access journal, articles about apps can get printed if the credit card clears.
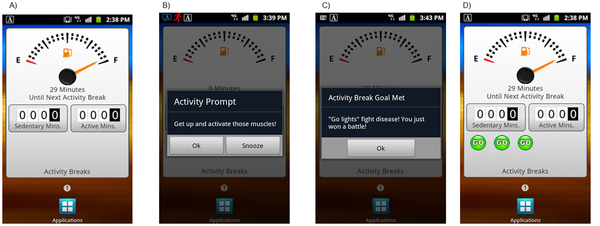
B-Mobile will remind you to stand up once in a while, though if you are obese because you sit and not because you eat too much pizza, you are in the minority.
doi:10.1371/journal.pone.0100821
Dale Bond, Ph.D., and Graham Thomas, Ph.D., lead researchers and faculty in the Department of Psychiatry and Human Behavior at The Miriam Hospital's Weight Control and Diabetes Research Center, worked with their colleagues to develop a smartphone-based intervention, or smartphone app, to reduce the amount of time obese individuals sit or recline while awake. The average American adult spends upward of 60 percent of his or her awake time being sedentary, and this low-cost intervention could be made accessible to a large segment of the population using a device they already own.
"Almost everyone knows that physical activity is important," said Dale Bond, Ph.D., at Miriam Hospital's Weight Control and Diabetes Research Center , "but it's not widely recognized that someone who runs five miles in the evening but spends the rest of the day sitting at a desk can be putting their health at risk. That smartphone you use so often throughout the day could now actually help to improve your health."
The smartphone app, "B-Mobile," was tested in a study of primarily middle-aged women who were obese, although the intervention can be applied to those who are not obese. The app automatically monitored the time participants spent being sedentary, and after an extended period with no activity, prompted participants via a tone paired with motivational messages to get up and walk around for a few minutes.
Participants received feedback providing encouragement for taking a break and reinforcement when they achieved the walking break goal. Researchers tested three different approaches to see which was best at reducing the total amount of sedentary time. Even though all three were successful, researchers found it is better to take shorter breaks more often for better health.
Also, while previous interventions have used similar behavioral strategies such as self monitoring and feedback to reduce sedentary behavior, use of a smartphone allowed these strategies to be easily automated and implemented through the day in any environment. The app performed better than other low-intensity intervention approaches that do not involve intensive face-to-face contact and/or expensive equipment.
"Prompting frequent, short activity breaks may be the most effective way to decrease excessive sedentary time and increase physical activity in individuals who are overweight or obese," Bond concluded. "Further investigations should determine whether these excessive sedentary time reductions can be maintained long term and impact sedentary-related health risks."





Comments