In this short series of articles about coal, engines and energy I am trying to show something of the history behind our current knowledge of heat, energy and thermodynamics. As discoveries were made about the nature of heat, improvements were made in the efficiency of engines. Investigations into the theoretical maximum efficiency of any heat engine led ultimately to the discovery of the laws of thermodynamics.
The laws of thermodynamics are of wide applicability to the understanding of how, through our profligate use of energy, we humans are having such a dramatic effect on our global environment.
Everyone knows that heat can produce motion. That it possesses vast motive-power no one can doubt, in these days when the steam-engine is everywhere so well known.Nicolas Léonard Sadi Carnot.
One of the tasks of the scientist and the science writer is to demonstrate the falsity of many 'facts' that 'everyone knows' and to explain the truth so that, one day, everyone may come to know the scientific facts. For example: 'everyone knows' that fire radiates heat and ice doesn't. But physics demonstrates to us that if we pass air over ice, and the air is colder than the ice, then the ice will get colder. It can only do this by transfer of heat into its environment.
Guillaume Amontons observed that the pressures of gases fell with a reduction in temperature. He speculated in 1702 that a sufficient reduction in temperature would lead to the complete disappearance of gas pressure. This concept of an absolute zero of temperature showed that our human sense of cold and hot does not allow us to directly perceive that an apparently cold object can act as a radiant heat source.
Pierre Prevost, a Swiss physicist, investigated the nature of heat. His 'theory of exchanges' , 1791, showed that a body emits and absorbs radiant energy at equal rates when it is in equilibrium with its surroundings. Another way of expressing that theorem is to say that if a body is not at the same temperature as its surroundings then there will be an exchange of heat: heat will travel from a relatively hot source to a relatively cold sink.
Every heat engine depends only on an exchange of heat: on having a source and a sink at different temperatures. The Stirling engine, for example, uses a permanently contained working fluid which is alternately heated and cooled.
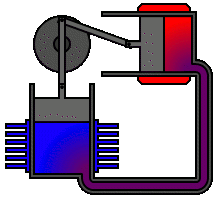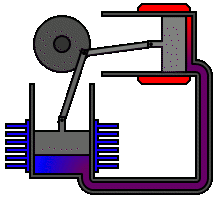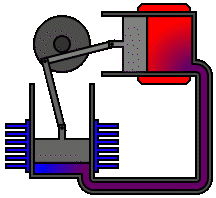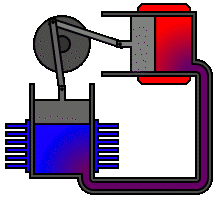
The Stirling engine affords a classic example of a heat engine that can make use of the relatively small differences in heat energy as, for example, between ice and air and between hot water and air. In the one case, ice, the air is a heat source. In the case of hot water, air is the heat sink:
It is quite within the realms of the laws of thermodynamics that we could, in theory at least, build engines which extract heat from ice and inject that heat into a colder environment. The power of such an engine would be just a matter of scale. Given a suitable environmental temperature and a sufficient quantity of ice at a higher temperature we could make the ice do the work of a horse.
Working Like A Horse
... an engine may be made large enough to do the work required in employing eight, ten, fifteen, or twenty horses to be constantly maintained and kept for doing such a work …James Watt quantified the classification of an engine according to how many horses it could replace. He used this as a means of determining the value for money - the cost/benefit - of his improved steam engines. Some figures on contemporary savings may be of interest:
Thomas Savery, 1702
In 1781, the wheel horses caught a break -- or were doomed, depending upon your point of view -- when James Watt patented the steam engine. In May of 1784, London's St. Katherine's Brewery replaced their four-horse mill with Watt's invention. The following month, Whitbread replaced its six-horse mill and was able to dispense with 24 horses who were costing them £40 each in upkeep per annum. (For £40, a lone engineer could be hired to tend the steam engine for an entire year.)A horse being, rather like a human, just another dumb animal whose rate of work varies with hunger, tiredness, reward offered etc., the value of a horsepower was differently estimated by different methods. It was eventually standardised somewhat arbitrarily as 33,000 foot pounds, i.e. the work done in raising a weight of 33,000 pound by one foot in one minute.
The Horse In Brewing History
Following the publication of An Experimental Enquiry Concerning the Source of the Heat which is Excited by Friction by Sir Benjamin Thompson, Count Rumford, in 1798, many scientists and engineers made enquiries into the nature of work, heat and energy. It gradually came to be accepted that heat is just one of many forms of energy and that different forms of energy can be converted one into the other. When all possible factors are considered, all energies can be accounted for. In the conversion of energy from one form to another there is no loss by the non-conversion or annihilation of energy. Energy is said to be conserved. In the second half of the nineteenth century the theory of the conservation of energy became an accepted 'given' of the physical sciences.
The theme of heat and energy is continued in Boring Science Produces Awesome Results
Related / further reading:
Biography Of James Watt
http://www.engineeringtoolbox.com/heat-work-energy-d_292.html
http://panda.unm.edu/Courses/Finley/P262/ThermalRad/ThermalRad.html





Comments