The first cases of Mad Cow disease in humans - properly known as variant Creutzfeld Jakob Disease, or vCJD, though no one recognizes that name - occurred in the late 1990s and were thought to be the consequence of eating contaminated beef products.
Since then, several cases of secondary infections caused by transfusions with blood from donors who subsequently developed vCJD have been reported, raising concerns about the safety of blood and blood products. A new paper describes an assay that can detect prions in blood samples from humans with vCJD and in animals at early stages of the (asymptomatic) incubation phase.
To develop the assay, Olivier Andréoletti, from the Ecole Nationale Vétérinaire de Toulouse, France, and colleagues first optimized a method called PMCA (for Protein Misfolding Cyclic Amplification). The method mimics in a test tube the process by which misfolded (toxic) prions propagate, and the researchers determined experimental conditions that enable efficient PMCA amplification of the vCJD agent in the blood.
Having defined such conditions, they show that the assay can detect vCJD in asymptomatic but infected animals in the early phase of the incubation period. They examined blood samples collected from infected sheep and macaques (vCJD-infected macaques are considered the best model of the human disease). In both models, the assay can accurately identify infected animals and detect the presence of vCJD prions in blood from macaques shortly after the initial infection (and several years before clinical disease onset).
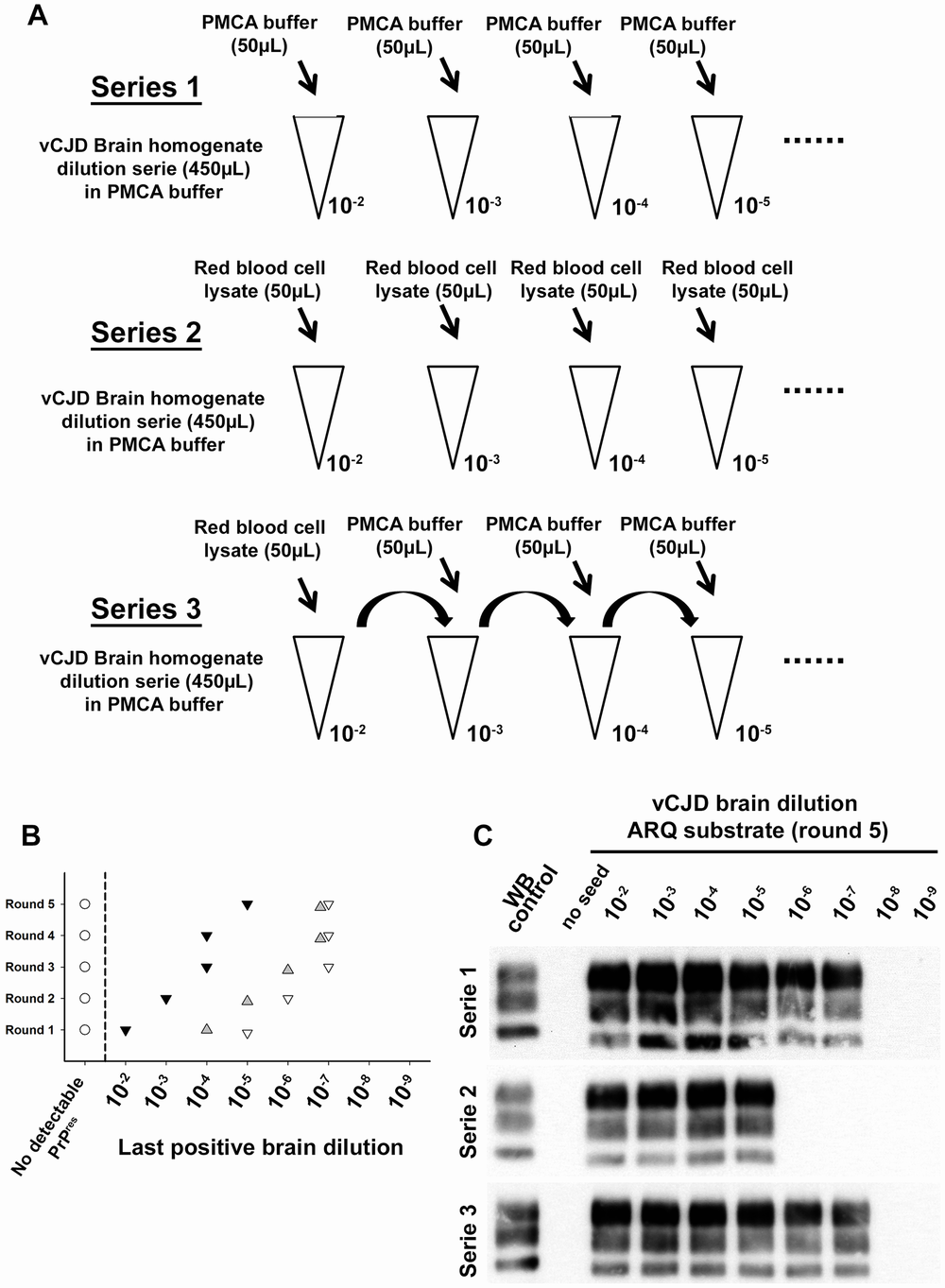
Red blood cell and inhibition of PMCA vCJD amplification. (A) Two vCJD brain homogenate (human) dilution series (1/10 dilution, 10−2 to 10−9) were prepared (aliquots of 450 µL). Each aliquot also contained 50 µL of PMCA buffer (series 1: ▿) or Red blood cell lysate (human) (series 2:▾). A third series (series 3: ) was prepared starting from a 10−2 dilution of the vCJD brain homogenate (900 µL) in which 100 µL of red blood cell lysate have been added. In each aliquot (450 µL) of dilution series 3, 50 µL of PMCA buffer were added. The three dilution series were then used to seed PMCA reactions (7 µL of seed) in which ovine PrP expressing mice (ARQ variant) was used as substrate (63 µL). Five successive rounds of PMCA were performed. After each round PrPres detection was carried out in PMCA reactions by Western blot (Sha31 anti PrP monoclonal antibody: epitope: YEDRYYRE, amino acid 145–152). Ten unseeded controls (○) were included in the experiment. The results of the PMCA amplification after each round are presented in graph (B). WB corresponding to the fifth round of amplification is presented as an illustration (C).
doi:10.1371/journal.ppat.1004202
Samples from human vCJD patients are rare, and none exist from individuals at preclinical stage of the disease. To test the assay in human blood, the researchers obtained samples from vCJD patients and non-infected controls and analyzed them blindly (i.e. the people who did the assays did not know which samples were which) in two different laboratories. The assay correctly and consistently identified three of the four vCJD affected patients, and yielded no false-positive result in any of the 141 healthy controls. The negative result in one of the vCJD samples raises the question of the potential absence of vCJD agent in the blood of certain patients.
The authors say their "results represent substantial progress towards an applicable vCJD blood detection assay. Such assay could be used to identify vCJD infected but asymptomatic individuals and/or for screening donated blood for the presence of the vCJD agent".





Comments