In the long term the ozone layer will recover thanks to extensive environmental policy measures enacted decades ago for its protection. This winter's likely record-breaking ozone loss does not alter this expectation.
Our atmosphere has five layers; the troposphere, the stratosphere, the mesosphere, the thermosphere, and the exosphere. The so-called ozone layer is in the stratosphere at an altitude of 20-30 kilometers. As you might infer, the ozone layer has high levels of ozone, which is due to interaction between oxygen molecules (composed of two oxygen atoms) and ultraviolet light. Ozone is able to absorb harmful ultraviolet radiation, wavelengths between 290-320 nanometers.
Ozone is lost in three ways: Volcanic eruptions in a small sense (5%), other natural occurrences (20%), but the bulk of the ozone breakdown happens when anthropogenic chlorofluorocarbons (CFCs) are turned into aggressive, ozone destroying substances - an effect made worse during exposure to extremely cold conditions.
For several years climate scientists have pointed to a connection between ozone loss and climate change, and particularly to the fact that in the Arctic stratosphere where the ozone layer is, the coldest winters seem to have been getting colder and are leading to larger ozone losses, though "the complicated details of the interactions between the ozone layer and climate change haven't been completely understood yet and are the subject of current research projects," explains Alfred Wegener Institute climate researcher Markus Rex. "The current winter is a continuation of this development, which may indeed be connected to global warming."
The Dobson Unit - how ozone is measured
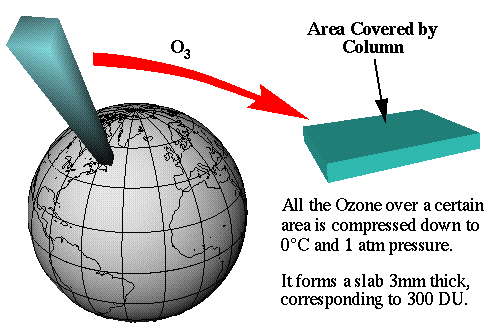
A Dobson Unit is a measurement of how thick the ozone layer would be if it were compressed into a single layer at zero degrees Celsius with one unit of atmospheric pressure acting on it (standard temperature and pressure - STP). One Dobson Unit (DU) is defined as .01 mm thickness at standard temperature and pressure. This graphic from the University of Michigan shows a column of air over Labrador, Canada. Since the ozone layer over this area would form a 3 mm thick slab, the measurement of the ozone over Labrador is 300 DU.
As stated before, this loss is not a reason to panic, though primarily because environmental changes were made before the situation was critical.
"By virtue of the long-term effect of the Montreal Protocol, significant ozone destruction will no longer occur during the second half of this century," explains Rex. The Montreal Protocol is an international treaty adopted under the UN umbrella in 1987 to protect the ozone layer and for all practical purposes bans the production of ozone-depleting chlorofluorocarbons (CFCs) worldwide today.
CFCs released during prior decades however, will not vanish from the atmosphere until many decades from now. Until that time the fate of the Arctic ozone layer essentially depends on the temperature in the stratosphere at an altitude of around 20 km and thus will linked to any changes in earth's climate.






Comments