The researchers found that p53 limits invasion by initiating a chain of events that ultimately prevents the formation of lamellipodia, cell membrane protrusions that spur cell movement and invasion. p53 activates a mitochondrial protease called Omi, which is then released into the cytosol of the cell when Ras causes mitochondria to fragment. Omi cleaves actin filaments in the cytoskeleton, and the decrease in actin suppresses the activity of p130Cas, a focal adhesion signaling protein that promotes the formation of lamellipodia. With low levels of active p130Cas, cells don't form lamellipodia and are therefore less able to invade.
"Actin remodeling is a signal that prevents cell invasion," explains Kawauchi. "Most research has focused on how p53 prevents metastasis by regulating epithelial-to-mesenchymal transitions," a biological process by which cells gain migratory and invasive properties. In contrast, says Kawauchi, the new findings help explain how p53 affects the cytoskeletal processes within the cell that drive invasion.
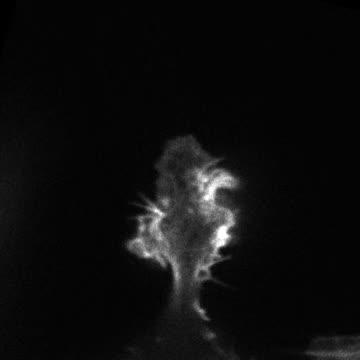
In the absence of Omi, Ras-transformed tumor cells form invasive lamellipodial protrusions.
(Photo Credit: Yamauchi et al., 2014)
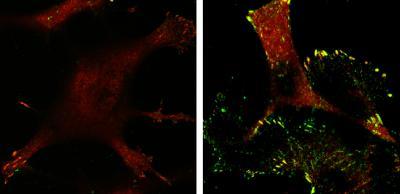
Tumor cells expressing p53 (left) have fewer focal adhesions (red) and lower levels of active p130Cas (green)—rendering them noninvasive—compared with p53-deficient tumor cells (right).
(Photo Credit: Yamauchi et al., 2014)






Comments