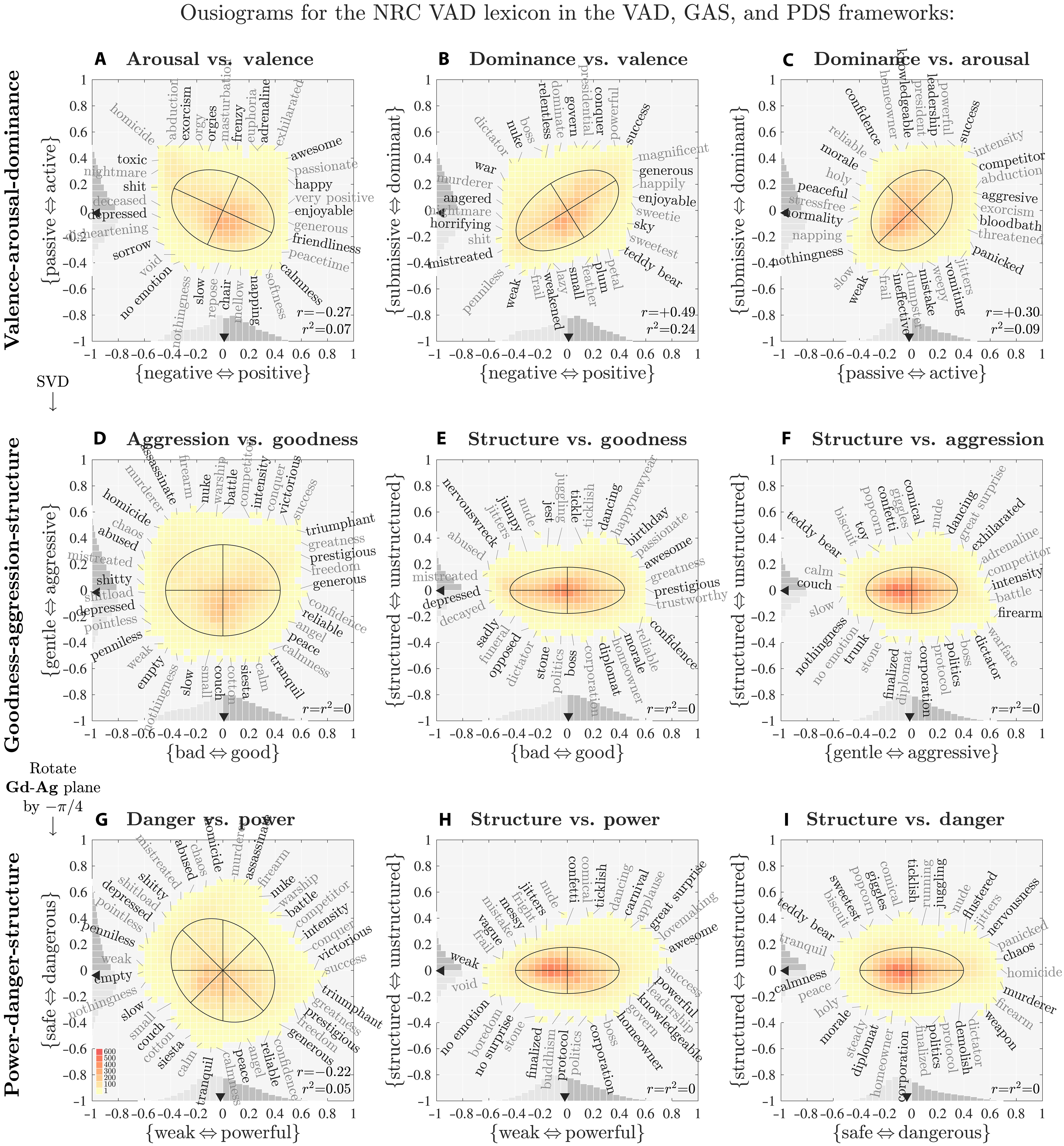
 Ousiometrics Analysis Says All Human Language Is Biased
Ousiometrics Analysis Says All Human Language Is Biased A new tool drawing on billions of uses of more than 20,000 words and diverse real-world texts claims...
 Wavelengths Of Light Are Why CO2 Cools The Upper Atmosphere But Warms Earth
Wavelengths Of Light Are Why CO2 Cools The Upper Atmosphere But Warms EarthThere are concerns about projected warming on the Earth’s surface and in the lower atmosphere...
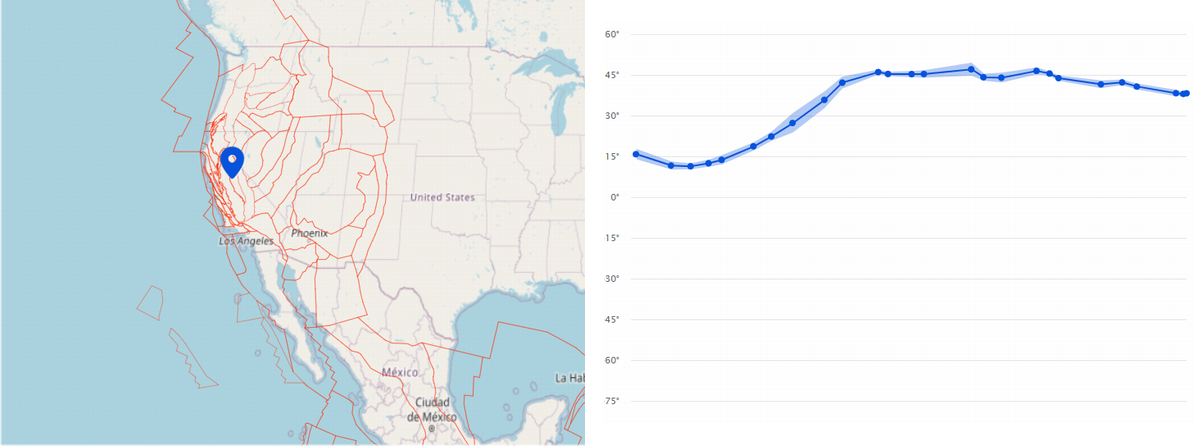 Here's Where Your Backyard Was 300 Million Years Ago
Here's Where Your Backyard Was 300 Million Years AgoWe may use terms like "grounded" and terra firma to mean stability and consistency but geology...
 Convergent Evolution Cheat Sheet Now 120 Million Years Old
Convergent Evolution Cheat Sheet Now 120 Million Years OldOne tenet of natural selection is a random walk of genes but nature may be more predictable than...