You Didn't Feel Continental Mantle Earthquakes, But They Happened. A Lot
You Didn't Feel Continental Mantle Earthquakes, But They Happened. A LotA 1979 seismic event was a different kind of earthquake, and it is has intrigued scientists ever...
 How To Overcome Leadership Battles
How To Overcome Leadership BattlesIn times of social rancor and strife, most will fight each other, but societies are saved by those...
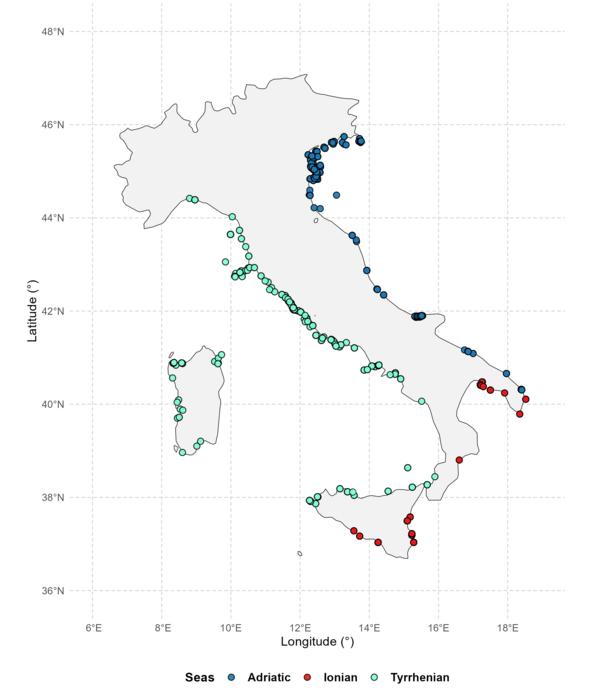 Thousands Of Unpublished Studies Show Why Conservation Efforts Miss The Mark
Thousands Of Unpublished Studies Show Why Conservation Efforts Miss The MarkEurope alone has so much unpublished, un-catalogued biological data that it is challenging to take...
 Why Antarctic Sea Ice Stopped Growing In 2015
Why Antarctic Sea Ice Stopped Growing In 2015Though numerical models and popular films like An Inconvenient Truth projected Arctic ice...