Every organism needs nitrogen to survive and grow and many organisms do not have the ability to obtain nitrogen from molecular nitrogen (N2), the major component in the atmosphere because they lack the nitrogen fixation pathway and have to rely on supply of nitrogen that has been fixed by others.
The availability of fixed nitrogen, in the form of ammonium, nitrite and nitrate, consequently often limits primary production in the environment. That's one of the reasons why many fertilizers are rich in fixed nitrogen.
There are microbial processes that convert fixed nitrogen back to N2 (production of energy instead of growth). Scientists call this process loss of fixed nitrogen, because it removes the important fixed nitrogen from the environment, and thereby limits primary productivity (i.e. production of biomass). These nitrogen-loss processes, which are carried out by different types of microbes, include the reduction of nitrogen compounds like nitrate and nitrite, the oxidation of ammonium, and a process that combines nitrite and ammonium to form N2, the anaerobic ammonium oxidation (anammox).
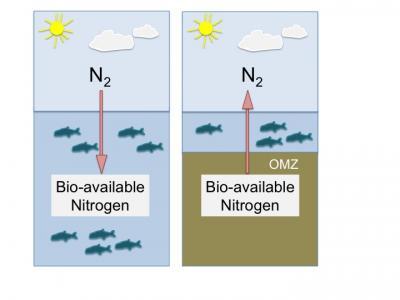
A schematic view of nitrogen net gain and loss in the ocean. Left: Nitrogen gas (N2) from the atmosphere diffuses into the ocean and microorganisms can fix this nitrogen into bio-available compounds, which are vital for every life form. Right: Situation in an oxygen minimum zone. If oxygen gets depleted, anaerobic processes, like anammox can convert fixed nitrogen to N2, which results in a loss from the pool of fixed bio-available nitrogen. The new study of Brunner et al addresses the different nitrogen-loss processes by using the isotope ratios for 15N and 14N in the pool for all nitrogen compounds. Credit: MPI for Marine Microbiology
In the water column of the ocean, these nitrogen-loss processes are most prominent in water bodies, known as oxygen minimum zones (OMZs), where dissolved oxygen (O2) is rapidly consumed almost to completion. There is major concern that such OMZs will expand in the future due to climate change, which could have a massive impact on the amount of nitrogen lost from the marine realm, affecting the primary productivity in the ocean.
For these reasons, it is important to know which microbial process is responsible for what part of the observed nitrogen-loss, and where this process happens within OMZs.
Co-author Ben Brunner of the Max Planck Institute for Marine Microbiology explains, "We can answer this question with the help of stable nitrogen isotopes, by looking at the ratio between the stable isotopes 15N and 14N in the different pools of fixed N and in the produced N2, because different microbial processes leave different N isotope fingerprints; some prefer the light isotope 14N over the heavier isotope 15N, and others do the opposite." Sergio Contreras, a (paleo) biogeochemist interested in the past and future of the Nitrogen cycling, continues: "However, the prerequisite to decipher the N isotope signatures in the environment is to know the isotope fingerprint of the individual nitrogen-loss processes".
Moritz Lehmann, isotope biogeochemist from the University of Basel, adds, "This is where so far, there was a gaping hole in our knowledge. The isotope effects of one major N-loss process, namely anammox, were unknown, and previous N-isotope based assessments of fixed N loss rates in the global ocean may have been severely biased."
Boran Kartal, microbiologist at Radboud University Nijmegen, explains, "We used the highly enriched cultures that are available in our laboratory to determine the nitrogen isotope effects of anammox bacteria. Our findings show that the isotope effects induced by anammox can explain isotope signatures observed in the OMZs, which are very important primary production sites in the oceans."
Marcel Kuypers, director at the Max Planck Institute, summarizes: "This missing piece of information is of utmost importance to solve the nitrogen isotope puzzle, not only because anammox is an important process in OMZs, but also because anammox simultaneously affects the nitrogen isotope composition of all nitrogen pools of interest: it converts ammonium and nitrite to N2 and nitrate."
Through their joint effort the scientists were able to decipher the intricate isotope fingerprint of anammox. Their results, published in the Proceeding of the National Academy of Sciences of the United States of America, reconcile so far mysterious N isotope patterns from OMZ, and provide the missing piece to solve the nitrogen isotope puzzle for fixed N-loss from the environment.






Comments