
One of the biggest struggles in toxicology is creating the correct parameters so you are modeling the real world as closely as possible. It's an enormous task to model the environment with its millions of factors, so controlled studies are done using animals.
Scientists design experiments that give an animal a lot of something at once and that can tell them 'this is the threshold where more analysis is a waste of time' and perhaps also find an effect that may be worth studying in more detail. It's a time-honored technique but it's also a technique that can be exploited.
Imagine you read a headline claiming that drinking Scotch was linked to massive brain damage and sudden death. That's an alarming finding and it's likely you would stop drinking Scotch due to the precautionary principle, even if you had consumed Scotch safely in the past.
But you would have a much different impression if the first sentence of the article noted that it would take 10,000 shots of Scotch to get that dangerous effect. You would no longer worry about having a Scotch before dinner and would pour a few ounces and start writing Congress asking why scientists wasted all that whisky on rodents who couldn't possibly have enjoyed it.
Drinking 10,000 shots of Scotch at once is not a real world possibility. Yet beyond the pale of reality is how a large number of toxicology results get portrayed - and the public doesn't know it unless they read the fine print.
*****
An Empty Scotch Barrel Makes The Loudest Noise - Scottish Proverb
*****
Toxicologists don't do studies on Scotch, of course, there are too many compounds to try and control for, so if they want to know about the effects of scotch they instead test if something specific in Scotch is harmful. To test for such effects, they will often pump in the chemical of interest using a technique called gavage (dosing) - a tube inserted into the stomach of the animal. To improve uptake of the chemical, they will use a surfactant; basically a detergent that breaks the natural surface tension of something, such as between a chemical and animal tissue. A famous surfactant experiment in K-12 science labs is a needle and water. When laid flat, a sewing needle will be held up by surface tension and float on top of water. If you add a little bit of detergent, however, that surfactant eliminates the surface tension and the needle immediately sinks.
Gavage dosing and surfactants are established techniques and in a prospective, well-controlled study they can speed up results and provide valuable information. But for people in the business of scaring the public, those techniques can easily be exploited to create a problem. It's not hard to do, because the techniques to create a desired effect, and the comparison to No Effect Levels in the real world, require expertise to find in science journal articles and so they rarely make their way into news coverage or press releases. Sometimes the data and methodology are not included in journal articles at all, making the claims even less credible.
You and I don't want to know what happens if we jam a tube of chemicals -- in this case, alcohol, but more commonly something like pesticides -- into our stomachs. We know that alcohol itself is constantly metabolized by our bodies so we are never storing 10,000 shots worth of alcohol all at once. What we would want to know is whether or not alcohol at small levels can build up over time and be harmful.
To find out where effects are, researchers test across ranges that are realistic, from 1 to 100 shots of Scotch, for example, 2 or even 3 of what are called orders of magnitude. An order of magnitude is when you multiply something times 10, as in 10, 100, 1,000, etc.
No one drinks more than 100 shots of Scotch at one time, not even on election night at Republican headquarters in 2012, so studying beyond that is unnecessary. But sometimes science does it anyway.

More than 10,000 shots of Scotch, in whisky barrels at Bruichladdich Distillery.
Photo by Meredith at Ponder the Irrelevant.
Let's talk about practical example of how dosing and method can impact results. In February of 2014, the herbicide atrazine made news when the New Yorker published a personal profile of a researcher, Professor Tyrone Hayes of U.C. Berkeley, who alleged that he was being persecuted by its manufacturer, the Swiss company Syngenta, for believing atrazine was harmful to frogs. His claims had been published in journals and though they included no data, the Environmental Protection Agency convened Scientific Assessment Panels (SAPs) anyway and after years of study the product was cleared. Regardless of the EPA finding no basis for concern, environmentalists advertised the claims of danger, without ever examining how valid the tests were.
By the second of these SAPs, held in 2010 and 2011, Syngenta had proactively done some follow-up work to clarify what the EPA really wanted to know - if there were effects in the real world. They decided to find out if it was even possible to get enough atrazine into the bodies of animals to have an effect. What they discovered should make people pretty comfortable about whether or not this pesticide can be harmful over time - at least as long as we don't have tubes and detergent being jammed into our stomachs.
Dr. Tim Pastoor, Principal Scientist at Syngenta, supervised studies of atrazine using conditions mirroring our environment - in other words, with atrazine dissolved in water, which is the only realistic way humans and animals can be exposed to it. At the same time, they decided to test the outer limits, going well beyond any realistic range of what people would actually ever be exposed to. So rather than using 2 or 3 orders of magnitude, such as 0.1 shots of Scotch to 100, they went all the way up to 5 orders of magnitude - 10,000 shots.
To put that range in context, 5 orders of magnitude is like the difference between the population of Canada and the population of 5 whole Earths. It's overkill to use 5 orders of magnitude, but the studies put an end to the atrazine controversy for the EPA - among other things, they showed that in the real world, you could not ingest enough atrazine to cause harm. The transcripts and data, all 1,000 pages from 2010, are publicly available, but I called Pastoor to talk about one particular slide he used at the public hearing:
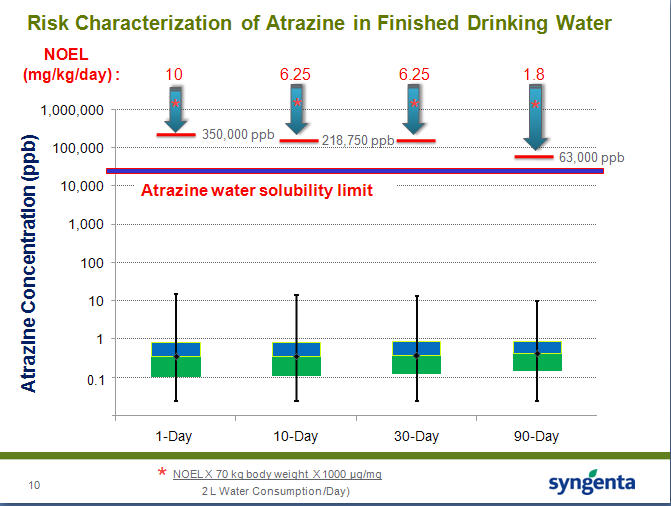
Atrazine levels in water compared to solubility in water compared to the EPA’s No-Effect Levels (NOELs) from animal testing, with water sampling timeframes. Source: Syngenta/Tim Pastoor via the EPA
NOEL is the No-Observed-Effect Level. In toxicology, that means an exposure level where there is no statistical or biological effect different than what is found in a control group - animals, in this case, who didn't get any exposure at all. The arrows with the parts per billion shows the no effect levels over the short and long term. Those are levels where there was no effect even using gavage dosing. Note that the vertical axis of the graph is exponential, increasing by a factor of ten between each line, so the distance between the lines as you go up the graph is much, much greater than it appears, sort of like how countries in the northern latitudes look so much bigger than countries on the equator in a flat map projection.
The line below that is the water solubility limit - how much atrazine can actually go into water.
We all remember in high school chemistry class where we learned that you can only add so much salt or sugar or anything else to water before it just clumps at the bottom. This is the same concept. At the bottom are the measured levels of atrazine found in actual water in numerous real-world water samples.
The study showed the actual levels of atrazine in water are pretty low and the levels the EPA worries about are way up at the top - and between them are the laws of physics. You can't actually dissolve enough atrazine in water to get a harmful effect.
What about toxic build-up over time, I asked Pastoor? The reason to use gavage dosing and surfactants is to try and simulate in a short amount of time what might be happening after build-up over many years. A month for a rat is about 30 of our months, so 90 days in their test was almost 8 years. Would it be any different if we tested for 25 years?
"We have done all of these other studies on looking at the mode of action for atrazine in mammals, we've done one of the largest kinetic studies ever done to determine how atrazine gets into the body, where it goes, what it does and so on. Typically, you'll take maybe 200 data points in a rat study to get your kinetics but in our study we had 1,800 data points to clearly delineate how atrazine entered the body, what the quantities were, what disappeared and went away," he said. "We found very clearly that atrazine enters the body, it is quickly metabolized and its metabolites are quickly excreted.
"We knew that if you gave it by a stomach tube you had different sorts of dosing than you did if you gave it by water or through the feed, as you might expect, but now we had the data to show that."
In their studies, they paired dosing up with actual effects and found that they could only get any effects by jamming the herbicide in all at once. If they fed the same high dose through water or the feed, they couldn't saturate it enough to get an effect. Even across 5 orders of magnitude.
"Right, if you're going to see it, you're going to see it across that extreme dose range," he said.
"On the basis of that, I came up with a statement that I own and I have said a million times; 'No one has, no one will and no one can get enough atrazine into them to cause an adverse effect.' That's an extreme way of saying that these minuscule amounts in water that you occasionally see are totally irrelevant to the biological system. Period."
"That's a bold statement," I cautioned.
He chuckled. "Most scientists will spend an hour explaining the kinetics and dynamics and the linkage with effects and no effects and you're just about ready to fall asleep and they say, 'on the basis of that, no one has, no one will'...so I'd rather start out with that and then explain why that is absolutely true. I think we get distracted by claims there is a science issue with atrazine and there's just not. It's a political story, and it's a litigation issue, and it's a self-aggrandizement story.
"But the actual science is boring because there's nothing going on."
Boring is good. Boring is what we want when it comes to pesticides, but is the issue ever really settled, regardless of the science? No, the EPA re-registers pesticides to address any new findings, that is why they exist, and atrazine is in re-registration again right now, just as it has has had to undergo re-registration periodically since it was first approved in 1959. The EPA monitors new studies because atrazine has been effective for over five decades and that means a lot of exposure.
Yet the laws of physics show that in normal usage, it can't come close to levels the EPA might consider harmful and, in real measurements, exposure is far below that. Professor Tyrone Hayes, the most vocal critic of atrazine and the scholar profiled in The New Yorker article I mentioned earlier, recently co-authored a paper finding that amphibian populations in the wild were not affected by atrazine exposure.
I'd be afraid to try and drink 10,000 shots of scotch. But I am not afraid of exposure in my water supply because 10,000 people might pour theirs down the sink. Understanding how chemicals work in the real world will make the public feel a lot better also.
*****
Top Scotch Credit: Andreas Levers on Flickr
NOTE:
(1) You will also find bits of strangeness. A representative from the Natural Resources Defense Council, Dr. Jennifer Sass, starts off her commentary with "again, the toxicology is really very expert, and you know that, and you've heard from the experts actually. So in a lot of them, I just say that I don't have an answer for that and I look forward to seeing what you guys come up with" and then after conceding it is not a human carcinogen still outlines that she has spent 9 years trying to get EPA to call it a carcinogen anyway.





Comments