 A New Target For Machado Joseph Disease Treatment
A New Target For Machado Joseph Disease TreatmentMachado-Josephdisease (MJD) is a hereditary neurodegenerative disorder that destroys the brainareas...
 Mechanism To Delay Aging Identified
Mechanism To Delay Aging IdentifiedAs medicine has improved, increasing our ability to treat disease, our longevity has improved as...
 New Treatment For The "Iron Overload Disease"
New Treatment For The "Iron Overload Disease"Hemochromatosis (HH) is the most common genetic disorder in the western world, and yet is barely...
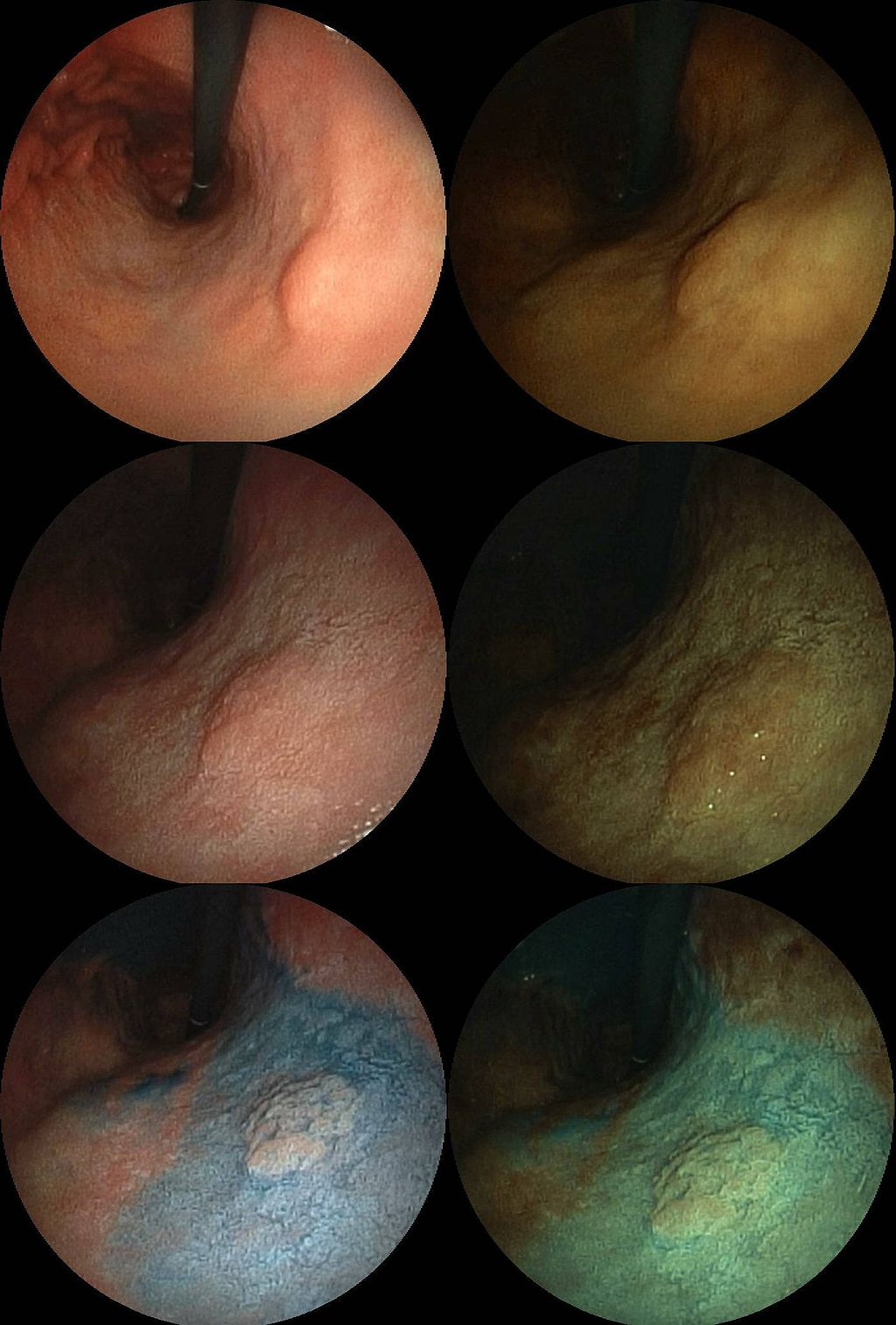
 Fluorescence Microscopy: New 2-D Images Can Detect Cancer Risk
Fluorescence Microscopy: New 2-D Images Can Detect Cancer RiskPortuguese researchers have developed a new method which uses images of a protein in cells to quantify...