Cancer is the blanket term for over a hundred diseases where abnormal cells divide and invade tissues through blood and lymph systems. The extra cells are often detected in the form of a tumor that people notice as their first symptom, and those can be benign or malignant.
A malignant tumor formed by cells that spreads outside the original tissue is termed a "metastatic" tumor. Because it is in addition to the primary cancer and has already spread, the danger is much higher - the 5-year survival rate for early-stage lung cancer is 52 percent while late-stage lung cancer that has metastasized to other areas of the body is just 4 percent.
What if metastasis could be halted? Researchers at Université Catholique de Louvain in Belgium have done just that, on models of human tumors in mice. Better yet, they say the prevention was complete - no recurrence.
That's right, they have cured this most dangerous form of cancer progression.
*****
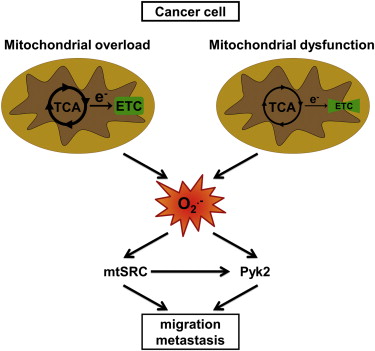
Mitochondria are the energy factories of cells. Aerobic glycolysis is the first stage in cellular respiration, the process cells use to release the energy stored in glucose. (See The Most Promising Antioxidant You Never Heard Of and Mitochondria And Antioxidants: A Tale Of Two Scientists for discussions of the other two stages, the Krebs cycle and the electron transport system). Cancers do have high aerobic glycolysis but defects in mitochondrial respiration have not been considered a cause of cancer.
Yet in their Cell Reports paper, the researchers identified protein tyrosine kinases Src and Pyk2, affecting mitochondria that overload the electron transport chain, leading to superoxide production. Learning that, they used a common research antioxidant, mitoTEMPO, to "scavenge" the mitochondrial superoxide and discovered that doing so blocked tumor cell migration and prevented spontaneous tumor metastasis in mouse and human tumor models.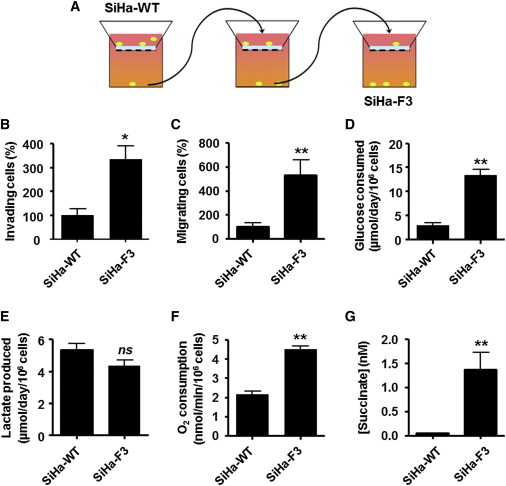
The Experimental Selection of Superinvasive Tumor Cells Correlates with Increased Mitochondrial Activity. (A–C) SiHa-F3 cells were isolated from a total population of WT SiHa tumor cells following three consecutive rounds of in vitro invasion in Matrigel-coated transwells (model shown in A). Comparative (B) invasion and (C) migration assays (n = 3). (D and E) Glucose consumption (D) and lactate production (E) over 24 hr (n = 3). (F) Mitochondrial oxygen consumption of adherent cells (n = 8). (G) Intracellular succinate levels (n = 5). All data represent means ± SEM.p < 0.05,p < 0.01, p < 0.005, ns, not significant compared to SiHa-WT by Student’s t test. Credit: DOI:10.1016/j.celrep.2014.06.043
In emails with senior author Professor Pierre Sonveaux, he said this could be the "Holy Grail" of metastatis research. To confirm the results and make them more relevant for potential human cancers, they also used MitoQ, because it has already gone through human clinical trials and so safety and dosing have been established. In both cases, Sonveaux said "targeting mitochondrial superoxide with inactivators such as mitoTEMPO and MitoQ, repressed metastatic progenitor cells and no metastasis was able to develop."What are the next steps? I asked. How many tumor models do you intend to study before you start calling venture capitalists and asking them to fund larger studies?
"The next step is to establish a strong cooperation with pharmaceutical companies owning inactivators of mitochondrial superoxides," Soveaux said. "Additional tests in mice are needed and are aimed to identify which types of metastatic tumors respond to the treatment, and which do not. In theory, the possibility exists that the vast majority of them will. Another important task is to evaluate the compounds in situations as close as possible to the clinics, with animals also receiving conventional anticancer treatments, such as chemotherapy and radiotherapy. At UCL, we have acquired a high level of expertise that, if the needed financial support is made available, will allow us to complete these tasks within 2-3 years. If both safety and efficacy are met, clinical trials would be launched."
Citation: Paolo E. Porporato, Valéry L. Payen, Jhudit Pérez-Escuredo, Christophe J. De Saedeleer, Pierre Danhier, Tamara Copetti, Suveera Dhup, Morgane Tardy, Thibaut Vazeille, Caroline Bouzin, Olivier Feron, Carine Michiels, Bernard Gallez, Pierre Sonveaux, 'A Mitochondrial Switch Promotes Tumor Metastasis', Cell ReportsCell Reports Volume 8, Issue 3, pp. 754–766, 7 August 2014. DOI:10.1016/j.celrep.2014.06.043





Comments